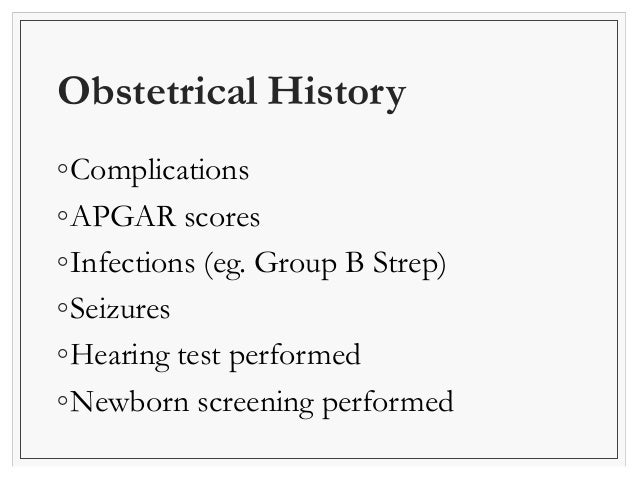
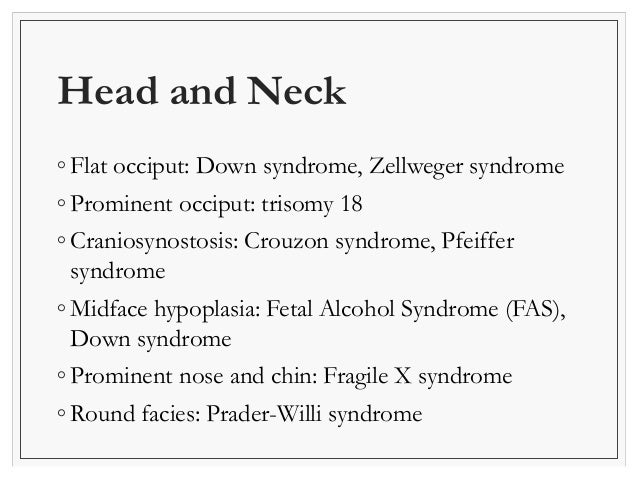
Newborn Examination Neonatology Videos-Click
PEDIATRICS – Study Material





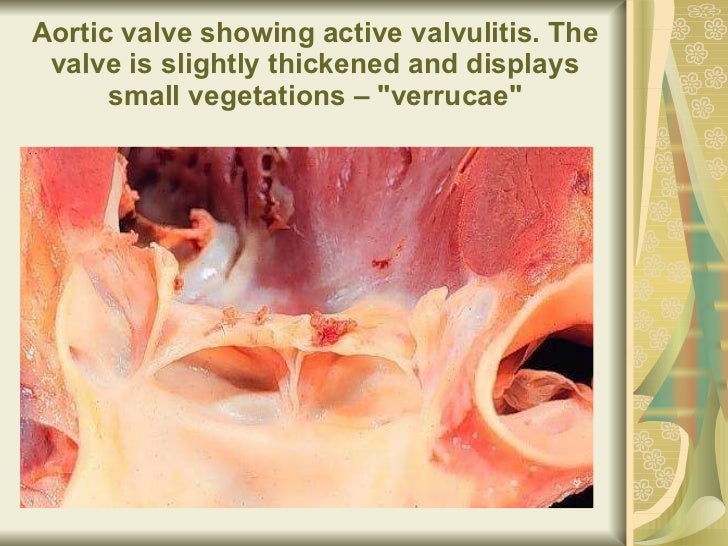




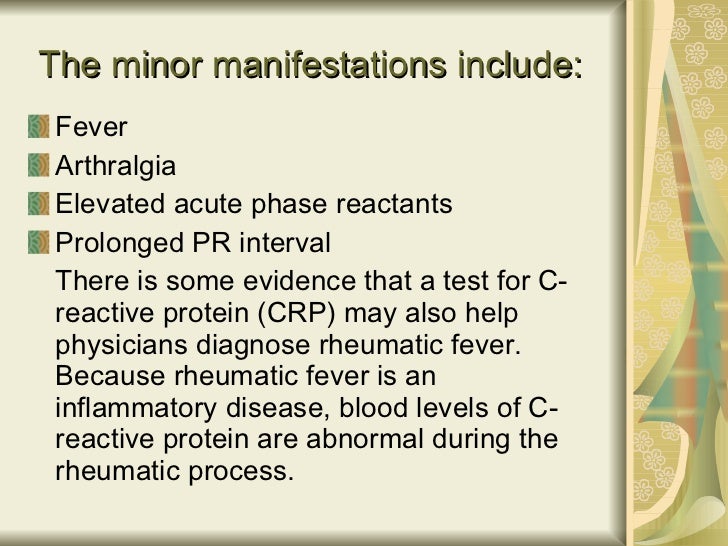

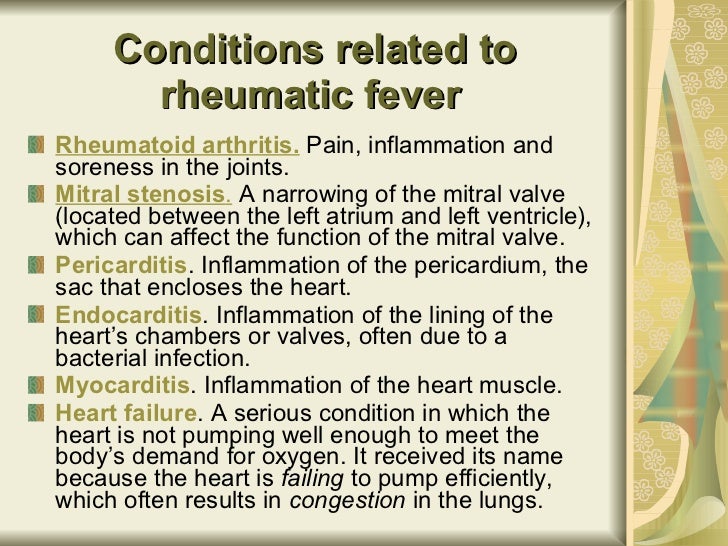



**********************************************************************************************************************************************************************




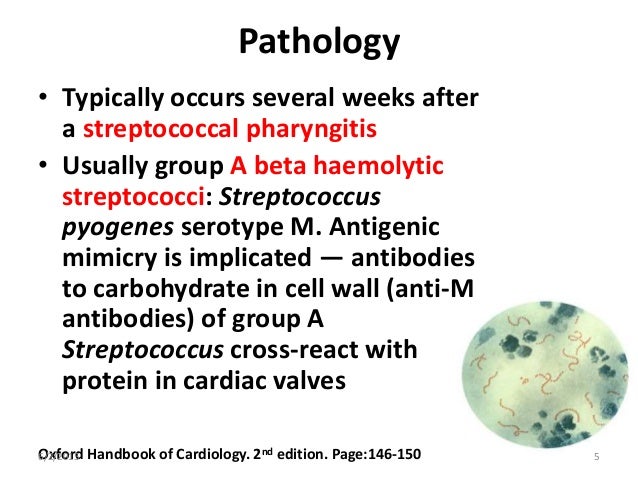













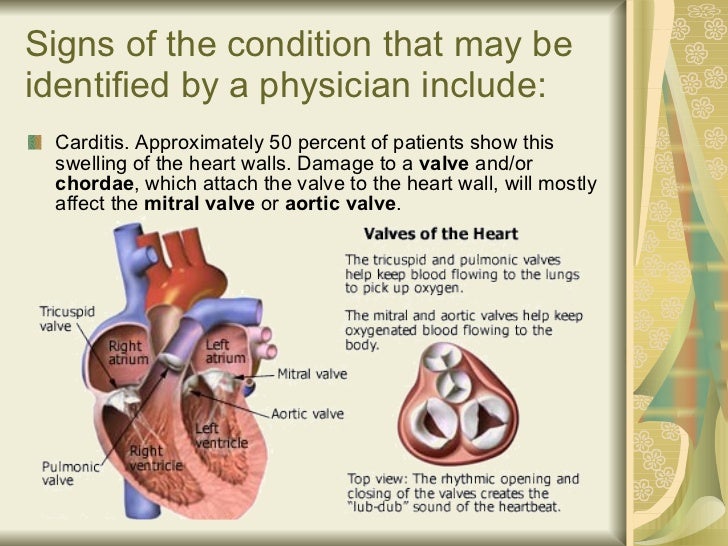
Cyanosis, or blue spells, is when a reduced amount of blood flows into the lungs. Since blood carries oxygen, less oxygen is delivered to the body. As a result, a child may appear blue or bluish. The colour is caused by high levels of reduced (deoxygenated) hemoglobin in the blood near the surface of the skin.
Cyanosis is a blue discoloration of the skin and mucus membranes caused by an increased concentration of reduced hemoglobin (>1.9–3.1 mmol/L) in the blood.
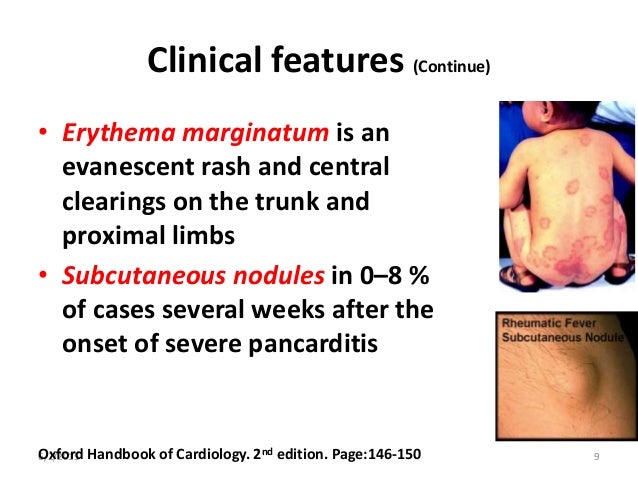
Peripheral and central forms of cyanosis are well recognized. Peripheral or acrocyanosis in newborns is regarded as a benign transient discoloration of the hands and feet. Central cyanosis is a serious pathological sign and involves discoloration of lips and tongue.
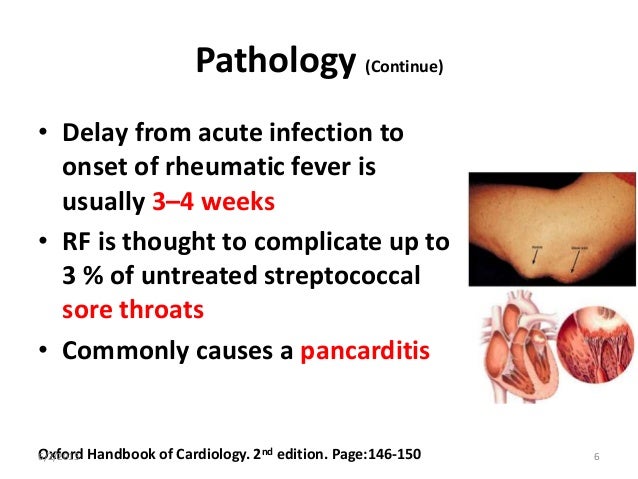
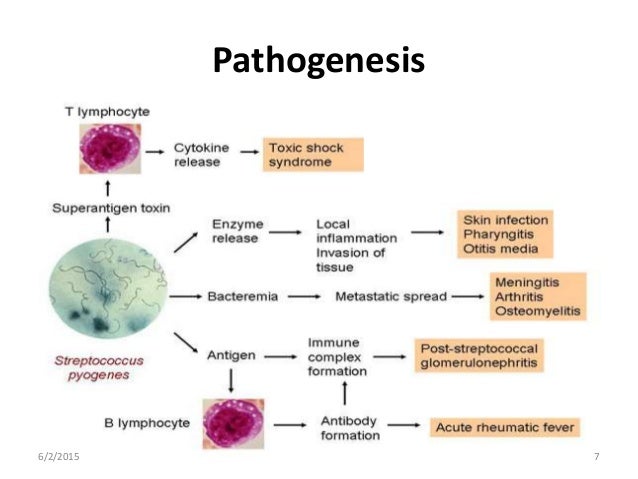
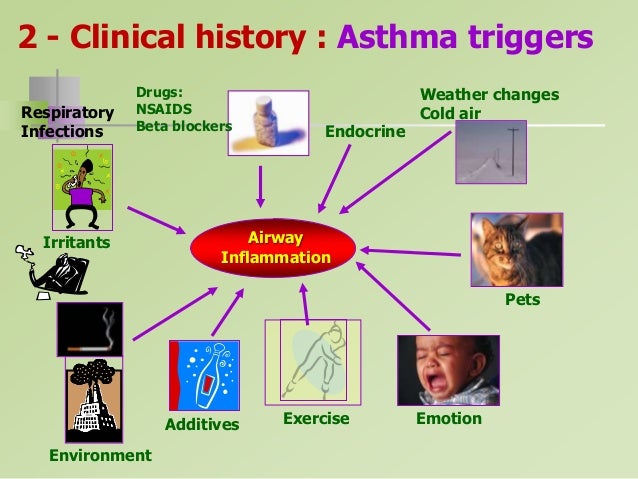
The list of the pathophysiological causes and the most common disorders summarized in Figure 1. Clearly, it can be a formidable task to reach the right diagnosis in a neonate with central cyanosis. The case presented below illustrates diagnostic and management challenges.

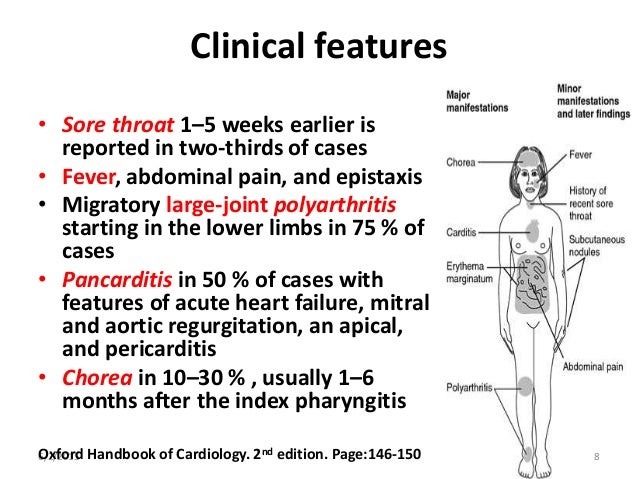
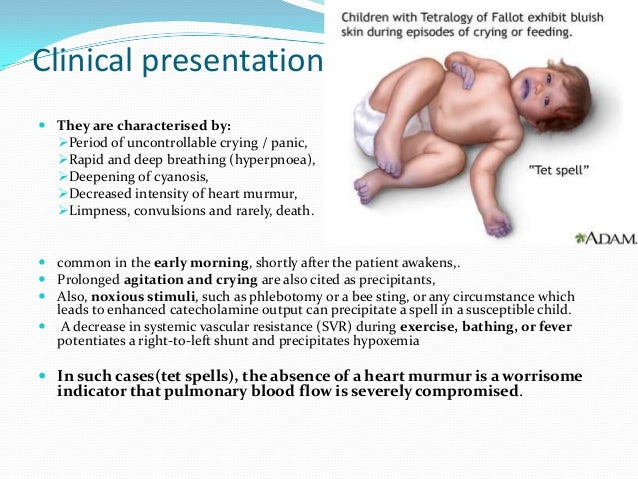
Cyanotic spells occur in children with cyanotic congenital heart disease, in particular tetralogy of Fallot and pulmonary atresia.
They usually occur early in the morning, or in the context of stress or dehydration ie. periods of increased oxygen demand/ultilisation.
The pathophysiology is not fully understood, but relates to decreased pulmonary blood flow.
Most episodes are self limiting.




















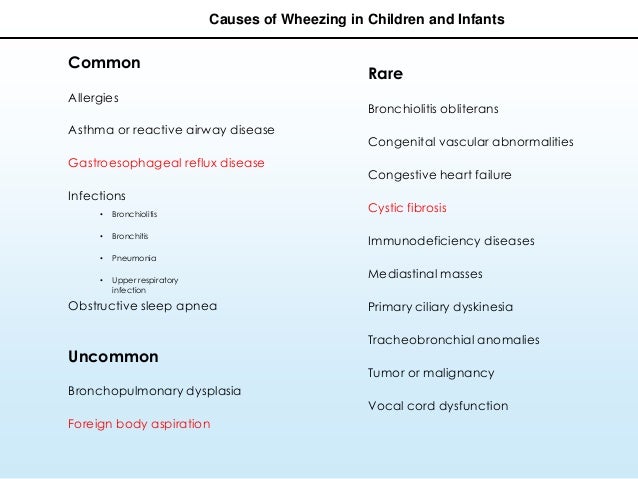
The most common causes of wheezing in children include asthma, allergies, infections, gastroesophageal reflux disease, and obstructive sleep apnea.
Less common causes include congenital abnormalities, foreign body aspiration, and cystic fibrosis.
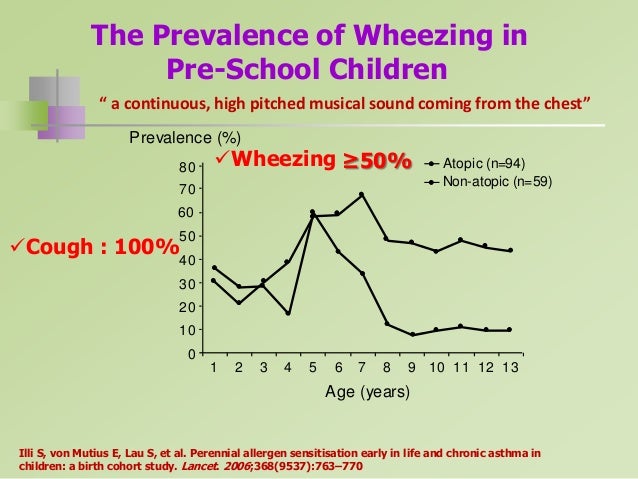
Recurrent wheezing have a significant morbidity and it’s estimated that about one third of school-age children manifest the symptom during the first 5 years of life.
Wheezing is a multi-factorial symptom, usually related to bronchiolitis or asthma.























































**********************************************************
Wheezing in Children Younger than 3: Differential Diagnosis and Initial Approach to Management
Wheezing is a common presenting complaint for the ED physician. The most frequent etiology of wheezing in childhood is asthma, which is estimated to account for 16.9% of annual ED visits by children.1 The evaluating physician must, however, consider a broad array of diagnoses in the wheezing pediatric patient. (See Table 1.)
This particularly is true when faced with the child younger than 3 years of age, who is more likely than the older child to be wheezing for the first time and for whom congenital anomalies gain relative importance in the differential diagnosis. The approach to the young, wheezing child begins with a thorough history and physical, that in many cases will narrow the differential diagnosis to several etiologies.
Appropriately selected studies further can elucidate the diagnosis. Regardless of the etiology, wheezing may be associated with respiratory distress, and the emergency medicine physician must be prepared to rapidly stabilize and provide definitive management of these children.
|
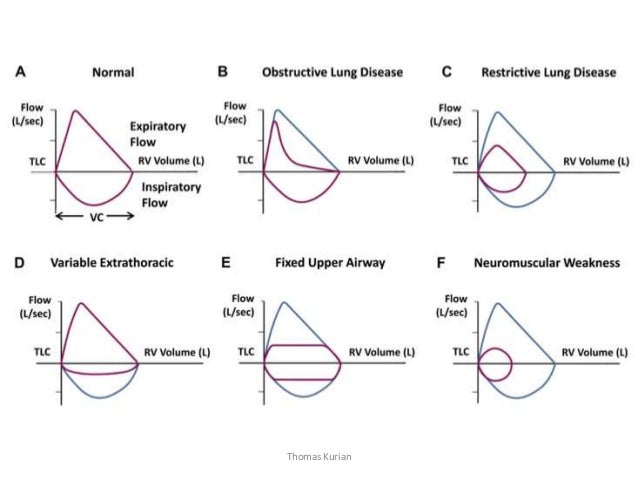
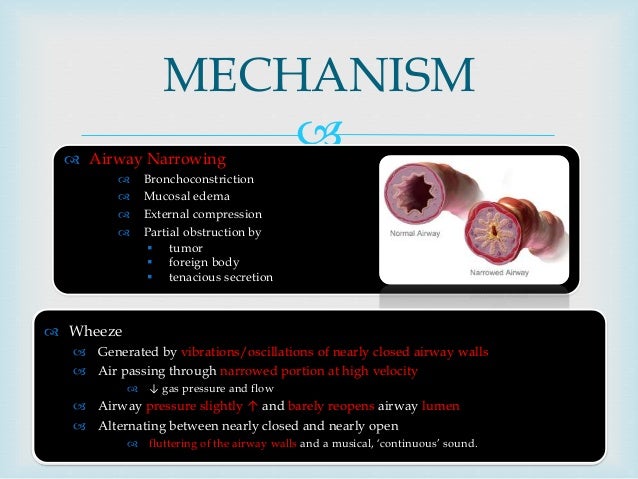
Pathophysiology
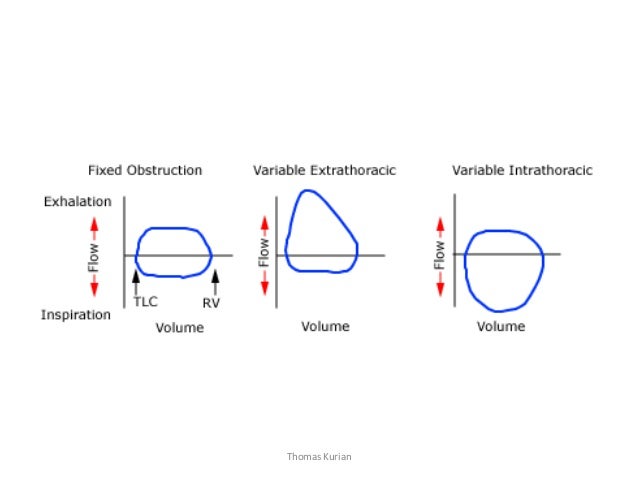
Wheezing is a high-pitched, continuous, adventitious lung sound. The net positive pressure in the chest during expiration compresses the intrathoracic airways; therefore, wheezing is initially an expiratory phenomenon. The wheezing sound is generated as the walls of the airways oscillate in response to the acceleration of gas flow through a narrowed lumen.2 Wheezing implies obstruction of the intrathoracic airways, and should be distinguished from stridor, a primarily inspiratory sound that occurs with diseases that affect the extrathoracic airway.3
Wheezes can be described as monophonic (sounding the same everywhere) or polyphonic (having a variety of pitches). Monophonic wheezes emanate from the large, central airways, while polyphonic wheezes are characteristic of small and medium-sized airway obstruction. Polyphonic wheezes are placed more peripherally and vary in pitch, depending on location.
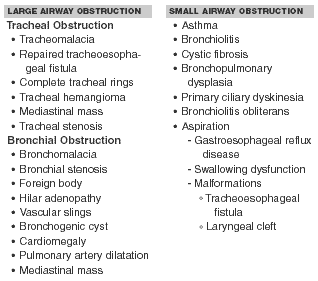
A common misconception is that wheezing occurs only with obstruction of the bronchioles and peripheral airways. However, obstruction of the intrathoracic trachea or bronchi also can result in wheezing because the expiratory compression forces are generated throughout the chest cavity.2 Intrathoracic airway diseases that result in wheezing can therefore be thought of as falling into two physiologically distinct groups: diseases of the large airways and diseases of the small airways.3,4 Uncomplicated large airway obstruction usually does not cause hypoxemia; the wheezing patient with a new oxygen requirement is likely to have a small airway component that induces hypoxemia through ventilation-perfusion mismatch. Table 2 differentiates common causes of large vs. small airway obstruction.
|
Airflow through the airway lumen may be impeded by intraluminal obstruction or extrinsic compression of the airway. In infants, intraluminal obstruction is more common. This intraluminal obstruction can result from bronchoconstriction, airway collapse, edema, or accumulation of mucus, depending upon the underlying disease.
Inflammation plays a central role in the development of intraluminal obstruction in many wheezing disorders, including asthma, infection, irritant or hypersensitivity reaction, and disorders related to aspiration. The release of inflammatory mediators leads to edema of the airway and bronchial smooth muscle contraction. Early mediators are those released from the mast cell, including histamine, leukotrienes, and platelet-activating factor, as well as proteases.5 Chemotactic factors, which recruit additional inflammatory cells to the site, include the interleukins, RANTES (regulated upon activation, normal T-cell expressed and secreted), and eotaxin.3 Activated helper T-cells in the airway release a variety of cytokines that enhance the inflammatory response. Late mediators are those released by inflammatory cells such as macrophages, lymphocytes, and eosinophils.
Infants and young children are at higher risk for obstruction of the tracheobronchial tree and wheezing than older children and adults.3 One reason for this is the small caliber of the infantile airway. Poiseuille’s law states that resistance to airflow varies inversely with the fourth power of the lumen radius. Thus, equal narrowing, whether by intraluminal or extraluminal factors, leads to a greater degree of resistance in the smaller airway of the young child.
Another factor that places infants and young children at increased risk for wheezing is the ultracompliant bronchial cartilage of their airways, which tends to collapse, resulting in premature closure of the intrathoracic airways with expiration. Additionally, histopathologic studies of infant lungs have revealed the absence of pores of Kohn and canals of Lambert.2 These structures, found in mature lungs, help maintain ventilation of the alveoli via collateral routes. The absence of these structures contributes to the susceptibility of infantile airways to obstruction and explains why apparently small alterations in an infant’s lung mechanics can lead to wheezing and respiratory distress.
Lung hyperinflation is a common finding in the wheezing, distressed infant or child. Increased airway resistance results in prolongation of the time necessary for the lung to empty. The tachypneic infant inspires before the lungs have returned to functional residual capacity (FRC), thus “stacking” breaths and resulting in air trapping. This air trapping leads to hyperinflation, which can be detected by physical examination and radiography.
Lung hyperinflation causes displacement of the diaphragm away from the chest wall. Though usually described as a “sheet of muscle,” the diaphragm more accurately is described as a piston. The costal fibers, which perform most of the work of the diaphragm, run parallel to the chest wall. With the diaphragm in its normal position, the net vector of force is parallel to the chest wall and the chest does not experience inward traction. However, hyperinflated lungs push the diaphragm away from the chest wall (a “flattened diaphragm”); when the costal fibers now shorten, they pull inward on their insertion at the bottom of the ribcage and cause a perceptible indrawing of the lower edge of the compliant ribcage of infants. In this situation, much of the diaphragmatic work goes to chest wall distortion and not to tidal ventilation. The infant diaphragm is more susceptible to fatigue than the diaphragm of an older child or adult, because the growth and development of the skeletal muscles of infants is incomplete, especially in infants born prematurely, and the proportion of fatigue-resistant fibers is less than that found in the adult diaphragm.6,7
Differential Diagnosis
Although the differential diagnosis of wheezing in young children is extensive, most wheezing in young children is attributable to asthma. (See Table 1.) The 1997 Expert Panel Report of the National Asthma Education and Prevention Program describes asthma as a chronic inflammatory disorder that presents with recurrent episodes of wheezing, breathlessness, and coughing, particularly at night or in the early morning; episodes are associated with variable degrees of airflow obstruction that is reversible spontaneously or with treatment.8 More than half of all cases of persistent asthma begin before the age of 3 years, and 80% have symptoms before the age of 5 years.9-11 Features consistent with asthma include the presence of an identifiable trigger such as upper respiratory infection (URI), a family history of asthma or allergies, and a response to bronchodilators and to corticosteroids.9
In infants and young children, the distinction between asthma and transient, virally induced wheezing may be difficult to make. The term “reactive airway disease” is sometimes used to refer to the collection of wheezing diseases in young children who are responsive to bronchodilator therapy, but for which a clear diagnosis of asthma has not yet been made. The term generally encompasses both atopic asthma and wheezing that is associated with viral illness. Further difficulty in distinguishing the entities occurs because viral infection is a frequent trigger for acute exacerbations of asthma both in children and adults. The presence of other atopic disease in the child or family, and evidence of sensitization to allergens, favors a diagnosis of asthma that is likely to persist. In most cases, children with recurrent wheezing have asthma; delay in making the diagnosis may lead to inadequate or inappropriate preventative therapy.
Bronchiolitis is a very common cause of wheezing in infants younger than 18 months of age; the peak incidence is between 1 month and 6 months. The etiology is viral, with respiratory syncytial virus (RSV) causing more than half of cases; other causes include parainfluenza virus and adenovirus.12-14 Symptoms result from obstruction of the small airways due to edema, accumulation of mucus, and bronchospasm.12 RSV-induced wheezing occurs most commonly in winter and early spring, and begins as a mild URI with rhinorrhea and sneezing. Fever is not uncommon early in the illness. Symptoms progress to involve the lower airways, with gradually worsening respiratory distress, wheezing, cough, irritability, and poor feeding. The physical examination typically reveals tachypnea with scattered fine rales and expiratory wheezing. Intercostal and subcostal retractions are common, and infants may be hypoxemic. Serious secondary bacterial infections are rare, but otitis media may occur. In very young infants, bronchiolitis may be complicated by apneic spells early in the course of the disease, particularly in children younger than 1 month. Chest radiographs, if done, usually reveal hyperinflation and scattered areas of atelectasis. The causative viral agent can be detected in nasopharyngeal secretions by antigen detection tests or by culture. Infants with underlying chronic illnesses, including congenital heart disease, bronchopulmonary dysplasia, immunodeficiency disorders, or cystic fibrosis (CF), are likely to have more serious illness than previously healthy term infants.12
Pneumonia in the infant or young child typically is a diffuse process that is viral in etiology.14 The most common etiologic agents are influenza, parainfluenza, and adenovirus. Classic lobar pneumonia, which usually is bacterial in origin, does not commonly cause wheezing.15 (See Figure 1.) Atypical organisms like Chlamydia pneumoniae and Mycoplasma pneumoniae may cause wheezing and diffuse pneumonia in school-age children, but these infections are uncommon in children younger than 3 years of age. Regardless of etiology, fever, cough, and rales often are more prominent findings than wheezing in children with pneumonia. An important etiology of pulmonary infection in very young infants is Chlamydia trachomatis. Infants acquire this infection perinatally from infected mothers, and may have a history of conjunctivitis within several weeks of birth. Up to 20% of perinatally infected infants develop pneumonia between 3 weeks and 3 months of age. Infection of the lower respiratory tract with C. trachomatis results in an afebrile pneumonia characterized by a repetitive “staccato” cough, tachypnea, and rales or occasionally wheezing.16
|
Environmental irritants such as tobacco smoke, smoke from heating sources, and fumes from cleaners and pesticides are an important and often overlooked cause of wheezing in infants and young children.17 Affected infants may display symptoms that wax and wane as they move between home and other environments. Environmental tobacco smoke exposure is not only a trigger for recurrent episodes of wheezing in predisposed infants and children, but also may be associated with an increased prevalence of asthma.18
Hypersensitivity is a more common cause of wheezing in adults than children, but may occur as a result of an IgE-mediated food allergy (most commonly to milk, soy, peanuts, fish, or shellfish), or in association with environmental molds or cockroach antigen. Symptoms typically occur immediately after exposure to the offending allergen. Hypersensitivity to an infectious agent occurs with visceral larva migrans and with allergic bronchopulmonary aspergillosis. Though hypersensitivity as a primary cause of wheezing is uncommon in young children, allergens frequently are implicated as the trigger for recurrent episodes of wheezing in young children with asthma and other chronic pulmonary diseases.
Aspiration of food, stomach contents, or foreign objects represents an important cause of wheezing in infants and young children. Respiratory symptoms associated with feeding and that began soon after birth suggest dysfunctional swallowing, gastroesophageal reflux disease (GERD), or rarely, an H-type tracheoesophageal fistula (TEF) or laryngeal cleft. Dysfunctional swallowing with associated aspiration occurs with increased frequency in children with developmental delays, neuromuscular abnormalities, and cleft palate. (See Figure 2.)
|
Of the many disorders associated with mucosal inflammation of the lower airway, GERD is one of the most common.19,20 GERD is thought to produce wheezing through two different mechanisms. Reflux of gastric contents into the esophagus leads to stimulation of vagal nerve endings in the distal esophagus, and results in cholinergic-mediated bronchoconstriction. Wheezing may also occur secondary to recurrent microaspiration of gastric contents, with resultant mucosal inflammation.9 Affected infants often have a history of spitting or emesis following feeds, and may be irritable because of chronic esophagitis. Many investigators report an increased incidence of GERD in patients with chronic wheezing, although it is difficult to decide whether GERD is the cause or consequence of wheezing.19,20 Aggressive treatment of GERD in patients with persistent wheezing often improves their respiratory status, suggesting that GERD at least partially is responsible for the wheezing.20
In the mobile infant or toddler, sudden onset of coughing, choking, or wheezing without associated viral symptoms should raise suspicion for foreign body aspiration (FBA), which is a common cause of morbidity and mortality in children between 18 months and 3 years.21-23 Inhalation of a foreign body into the respiratory tract may result in an acute life-threatening obstruction. More than 300 deaths per year in the United States occur as a result of FBA. Undiagnosed, retained foreign bodies may cause serious complications, including pneumonia, wheezing, bronchiectasis, and atelectasis. A high index of suspicion generally is required to avoid significant morbidity and mortality. The symptoms and signs can be confused with those of asthma, and the roentgenographic findings with those of asthma or pneumonia. Common clinical features of FBA are listed in Table 3. The triad of wheezing, paroxysmal cough, and decreased air entry, considered to be highly suggestive of FBA, is seen in only 35-40% of cases.21
|
Nuts are the most frequently inhaled foreign bodies in the United States, but seeds, including watermelon and sunflower seeds, also commonly are reported.21,24 Aspiration of an organic foreign body typically results in a worse clinical outcome than aspiration of an inorganic foreign body for a number of reasons. The elapsed time from aspiration to diagnosis is significantly shorter for children with inorganic vs. organic FBA. Aspirated organic foreign bodies may swell with bronchial secretions causing increasing obstruction, and they are known to initiate surrounding tissue reactions, which may cause inflammatory changes in the airways.23 During bronchoscopy, organic foreign bodies may be dispersed as smaller fragments in distal branches of the airway.24,25 One-fourth of patients who are not diagnosed within the first 24 hours develop respiratory symptoms such as cough or wheezing that are persistent following bronchoscopic removal of the organic foreign body. Duration of symptoms varies from 1 month to 3 years.24
Tracheomalacia and bronchomalacia are disorders commonly seen in tertiary pediatric respiratory practice.26-28 These disorders are characterized by a defect in the amount or composition of the airway cartilage. The affected segment(s) of the airway tend to collapse during exhalation, resulting in wheezing and air trapping. The defect can be congenital, or acquired from an infectious, mechanical, or other insult to the airway.4 The lesion often is diffuse and best described as tracheobronchomalacia (TBM).
These disorders of the large airways are associated with varying combinations of cough, wheeze, and stridor. Patients with malacia disorders tend to be noisy breathers early in life and have prominent chest congestion with colds. The parents of patients with tracheomalacia and bronchomalacia often describe their infants as being “born with a cold,” because the infants have chronic airway noisiness that mimics the congestion heard in URIs.
The spectrum of disease in tracheomalacia and bronchomalacia ranges from that which is life-threatening and requires major diagnostic and surgical intervention to mild cases that can be managed with observation and parental reassurance.26,27 The typical course for an infant with TBM is gradual improvement over time, with complete resolution of symptoms between the ages of 1 and 5 years. Rarely, newborns or infants with TBM will develop a life-threatening pattern of “dying spells” in which complete airway obstruction takes place. These infants require intubation and positive pressure ventilation.3
Important chronic lung diseases that present as wheezing in infancy and early childhood include bronchopulmonary dysplasia (BPD) and CF. BPD typically occurs in premature infants with prolonged exposure to mechanical ventilation and oxygen.29 Symptoms are exacerbated during viral infections. The disease gradually improves with increasing age and size of the infant. CF may present as wheezing and/or tachypnea that is persistent or recurrent in infancy. The presentation of CF is extremely variable and may include respiratory symptoms, poor growth, recurrent infections, or gastrointestinal symptoms with malabsorption.
Infants with recurrent episodes of wheezing in association with pneumonia, sinusitis, or other infections may have an immunodeficiency. Considerations include human immunodeficiency virus (HIV), humoral immunodeficiencies, and ciliary dyskinesia (the immotile cilia syndrome).
Although airway narrowing related to intraluminal obstruction is the most frequent cause of wheezing in infants and young children, extrinsic compression of the airways by adjacent structures requires mention. Important sources of extrinsic compression include vascular malformations, mediastinal masses, and cardiac disease. The specific source can be acquired, or may be present at birth.
Vascular malformations are potentially life-threatening causes of wheezing in infancy. An aberrant innominate/subclavian artery is a common vascular anomaly resulting in airway compression.30,31 Malformation of the aortic arch can result in a complete or partial ring that constricts the trachea.32 A double aortic arch is the most common type of complete vascular ring. Infants with vascular rings usually present with respiratory symptoms in the first six months of life.
Extraluminal intrathoracic airway narrowing may result from mediastinal sources.33 Masses that occur in the mediastinum of infants and children include a heterogeneous group of congenital, neoplastic, and infectious causes. (See Table 4.)
|
The mediastinum is divided into anterior, middle, and posterior compartments; most masses arise from normal mediastinal structures within a compartment, and there is a remarkable predilection for the mass to remain localize in the compartment of the structure from which it arose. Therefore, when a mediastinal mass is identified, localization to one of the three compartments aids in the differential diagnosis. (See Figures 3a and 3b: Chest radiographs of a child who presented with stridor and was diagnosed with a mediastinal mass.)33
|
|
The vast majority of children with mediastinal masses are symptomatic, and the frequency of symptoms is increased in children younger than 2 years and in those with malignant tumors. The clinical presentation is related to the patient’s age and the nature and anatomic location of the mass. Cough, respiratory distress, and neurologic manifestations are among the most commonly reported symptoms. In patients who are asymptomatic, many of the lesions are discovered incidentally during radiographic evaluation for other purposes.
Cardiac disease is an uncommon but important cause of wheezing in infancy. Wheezing may be the presenting symptom of congenital heart disease for infants with congestive heart failure, or may occur in association with cardiomegaly because of external compression of the airways by an enlarged heart. The infant usually has other signs of cardiac dysfunction, such as sweating, pallor, difficulty with feedings, and poor weight gain. In addition to wheezing, the physical examination may reveal crackles, a heart murmur, and hepatosplenomegaly. Cardiomegaly may be demonstrated with chest radiography. (See Figure 4.) The definitive diagnosis, however, usually results from echocardiography (See Figure 5.).34
|
|
Clinical Features
A careful history and physical examination can provide clues to the most likely diagnoses and clarify priorities for evaluation. Characteristic clinical findings for selected diagnoses in the differential are presented in Table 5.
• Clarify the age at onset and the pattern of wheezing. Recurring or persistent wheezing starting at or soon after birth suggests a diagnosis other than asthma or bronchiolitis; considerations include congenital abnormalities, perinatal infections, CF, or chronic lung disease of prematurity (e.g., BPD).9,35,36 Congenital abnormalities affecting the respiratory tract include TBM, TEF, vascular rings, airway stenosis, and webs. Wheezing associated with congenital heart defects also frequently presents in the newborn period, especially in children younger than 8 weeks of age. Recurrent aspiration from GERD or dysfunctional swallowing also might present at this time. Wheezing from CF may be present from birth and most commonly persists during the first year of life.36 Wheezing associated with prematurity and BPD usually is present at discharge from the nursery, and gradually improves with age and size, though infants may have worsening symptoms associated with viral infections, especially in the first year of life. Viral bronchiolitis is most common in the first year of life, and is especially common in infants 1-6 months of age who present with a first episode of wheezing. Wheezing from bronchiolitis rarely lasts longer than four weeks. Sudden onset of first-time wheezing in an older infant or toddler should prompt investigation for aspiration of a foreign body.
Recurrent episodes of wheezing in the first 3 years may represent multiple isolated episodes of virally mediated bronchospasm, or may herald a diagnosis of asthma. An attempt should be made to distinguish between recurrent episodes of wheezing triggered by repeated acute URIs, and chronic wheezing associated with underlying pathology. Examinations of previous ED records over time, including during times when the child is asymptomatic, may clarify the severity and chronicity of lung disease.36
• Identify other pulmonary complaints besides wheezing. Cough may be a more prominent symptom than wheeze with aspiration syndromes, including FBA, as well as with chlamydial pneumonia and CF.35 The presence of inspiratory stridor suggests an upper airway extrathoracic component of obstruction and is often noted with laryngomalacia, TBM, or a vascular ring.
• Investigate the association of symptoms with feeding. An infant with congestive heart failure becomes increasingly breathless or tires easily when fed. Infants with airway abnormalities such as TBM or a vascular ring may have worsening stridor or wheeze during a feed. Coughing, choking, or noisy breathing during or soon after a feed suggests aspiration from dysfunctional swallowing, a TEF, or GERD. Infants with GERD also may have a history of frequent emesis or irritability, especially after feeding or when laid down. Wheezing after food ingestion may also be a clue to allergen-induced food allergy, which can occur with or without gastrointestinal or skin symptoms.37
• Ask about symptoms of current or recurrent infection. Recent symptoms of a URI in an infant with a first-time wheeze suggests bronchiolitis; URI also may trigger recurrent wheezing in children with asthma. The presence of fever suggests an acute infection; viral infection is more common than bacterial in this age group, but fever should prompt a search for a coexisting bacterial infection. In the majority of cases, a coexisting bacterial disease will not be identified. Fever may accompany bronchiolitis or pneumonia, but absence of rales makes viral or bacterial pneumonia less likely. A history of recurrent pneumonia or other infections should raise a concern for immunodeficiency diseases, including HIV. Children with recurrent pneumonia also should be evaluated, initially by history and physical exam, for recurrent aspiration.38 A history of neonatal conjunctivitis increases the possibility of perinatal infection with chlamydia.
• Identify the trigger for the current episode of distress, especially for children with a known history of asthma or recurrent wheezing. URI is the most common precipitant of wheezing in children younger than 5 years.8 Environmental factors that may trigger wheezing include environmental tobacco smoke, fumes from household cleaners, airborne allergens (e.g., animal dander, molds, cockroach antigen, dust mites, and pollen), wood burning stoves, unvented heaters, and pesticides. Exercise, which may include giggling or crying in the young child, can exacerbate wheezing; children with exercise-induced worsening of symptoms are more likely to have asthma than other causes of wheezing.37
• Review the family history. Children with asthma often are found to have close relatives with asthma, allergic rhinitis, eczema, or recurrent sinusitis. CF is inherited as an autosomal recessive trait.
• Begin the physical examination by evaluating for signs of respiratory distress. Indications of respiratory distress include use of accessory muscles, chest wall retractions, nasal flaring, paradoxical breathing, cyanosis, respiratory rate in excess of 60, and oxygen saturation less than 91%.8
• Listen to breath sounds. The quality of air entry and the inspiratory to expiratory ratio (I:E ratio) should be assessed. The normal I:E ratio is 2:1 or 3:1; extrathoracic causes of obstruction may result in prolonged inspiration and inspiratory stridor, while intrathoracic obstruction is more likely to produce wheezing, prolonged expiration, and use of accessory muscles.38 When bronchiolar obstruction is almost complete, wheezing and breath sounds may become inaudible. Asymmetry of breath sounds may be a clue to consolidation or atelectasis. The presence of rales may imply pneumonia, CF, BPD, or pulmonary edema.35
For children with respiratory distress, initiate therapy, then complete the physical exam. Additional clues from the physical exam include:
Diagnostic Studies
Pulse Oximetry. Pulse oximetry should be obtained on all wheezing children; hypoxemia cannot be recognized adequately by clinical examination alone. An accurate pulse oximetry reading requires adequate peripheral perfusion with pulsatile blood flow. A reliable tracing is important and may be challenging to obtain, especially in younger children.
Blood Gas Analysis. Blood gas exchange is evaluated most accurately by the direct measurement of arterial partial pressure of oxygen (PO2), partial pressure of carbon dioxide (PCO2), and pH. Noninvasive monitoring with pulse oximetry has eliminated the need for blood gas analysis in some circumstances, but pulse oximetry does not provide information about ventilation. Blood gas analysis is necessary to evaluate PCO2 in patients with suspected hypoventilation or severe distress.8 Except in neonates, a PO2 less than 85 mmHg is abnormal in a child breathing room air; arterial PCO2 greater than 45 mmHg usually indicates hypoventilation or a severe ventilation-perfusion mismatch, unless it results from respiratory compensation for metabolic alkalosis.38 Care is necessary in interpreting the PCO2 in a tachypneic child. Hyperventilation associated with respiratory distress should result in a respiratory alkalosis; a “normal” or rising PCO2 in a tachypneic infant, especially when associated with acidosis, suggests significant respiratory compromise. Children with chronic lung diseases like BPD require special consideration. Over time, these children may develop a compensated state with elevated PCO2 balanced by an increased serum value of bicarbonate; respiratory deterioration is reflected by a rise of PCO2 above the child’s baseline or in association with acidosis.39
Pulmonary Function Testing. Tests of pulmonary function are useful in defining the type of pulmonary process (obstructive vs restrictive), the degree of impairment, and changes during treatment.32 Bedside measurement of expiratory flow rate has become standard in the older child with pulmonary disease. However, devices for pulmonary function assessment require cooperation and are not useful outside of research settings in children younger than 3 years old.
Chest Radiograph. Though chest radiographs have been advocated in the past for all children who present with wheezing for the first time, the utility of this study currently is in question. Although chest radiographs may provide clues helpful in diagnosing recurrent aspiration, CF, foreign body aspiration, pneumonia (See Figure 6), congestive heart failure, vascular ring with a right-sided aortic arch, or a mediastinal mass,37 the overwhelming majority of children with first-time wheezing have asthma or bronchiolitis.
|
Chest radiographs in these children may be normal or may reveal hyperinflation, peribronchial cuffing or thickening, interstitial infiltrates, or areas of atelectasis—findings that do not alter management.40 Prospective studies are limited; retrospective studies and case series suggest that radiographs can be performed in a selected population of wheezing children.41-44 Chest radiographs should be considered for an infant or child who presents with wheezing and any of the following:
Diagnostic imaging also is indicated for newborns with respiratory distress, and for infants or children with chronic cough or neurologic symptoms. When chest radiographs are ordered, both anteroposterior and lateral views should be obtained.
Imaging for children with suspected FBA deserves special comment. Most foreign bodies are radiolucent and unlikely to be evident on routine chest radiographs. Paired inspiratory and expiratory chest films (See Figures 7a and 7b) may reveal an area of lung which fails to empty appropriately with expiration, or a shift of the mediastinum away from the side with an obstructing foreign body; however, these films may be difficult to obtain in the uncooperative toddler. Bilateral decubitus chest films may reveal failure of the dependent lung to deflate.9 Observation of respiration under fluoroscopy is a particularly sensitive method of evaluating for abnormal movement of the diaphragm or mediastinum that may occur in the presence of a foreign body, but this study may not be widely available. Bronchoscopy is indicated whenever the suspicion for a foreign body is high, even if the radiographs are normal.
|
|||||||||
Viral Studies. Efforts to identify the viral etiologic agent in bronchiolitis and pneumonia generally are unnecessary. Identification of a specific organism may be useful for isolation and cohorting of hospitalized children. Tests for C. trachomatis should be obtained in appropriate clinical settings, since the diagnosis requires systemic treatment not only of the infected infant, but also of the mother and her contacts.
Other studies which may be useful in diagnosing specific entities are listed in Table 5.
Management—General Principles
• Begin with stabilization of the respiratory status. In the child in respiratory distress, therapy should be initiated after a brief, focused history and exam; a more detailed evaluation including laboratory or radiologic studies should be done only after therapy has been started.
• Ensure a patent airway. Alert children should be allowed to determine their own position of comfort. The airway should be cleared of secretions; this is particularly important for infants with bronchiolitis. Humidification is helpful in preventing obstruction of small airways by dried sections.39 Children with significant respiratory distress should not be given anything to eat or drink (NPO).12
• Provide oxygen and monitor oxygen saturation.8 Most children with wheezing and any degree of respiratory distress will benefit from oxygen, even if pulse oximetry values are normal.39 Oxygen should be provided in a nonthreatening manner, since anxiety and agitation increase metabolic demand and oxygen consumption. Alert children with respiratory distress should be allowed to remain with their parents, who may be able to position oxygen on the child’s face or blow it near the nose and mouth without causing agitation.
• Provide a trial of beta-agonist therapy. Many, though not all, children with wheezing will benefit from the bronchodilating effect of a beta-agonist such as albuterol. Exceptions include infants with bronchiolitis, who typically show no response to bronchodilating medications, and children with wheezing associated with a suspected cardiac lesion, for whom beta-agonist therapy is contraindicated.12,13 The patient should be evaluated carefully prior to the administration of the beta-agonist medication and shortly following its completion. Young infants who show no improvement should not be given additional doses. For patients who demonstrate improvement with beta-agonist therapy and persistent respiratory distress, the medication may be repeated every 20-30 minutes until the child has resolution of symptoms or is clinically breathing comfortably. See Table 6 for drug dosages.
The best method for delivering beta-agonist therapy has been studied extensively. Nebulizers do not require coordination and are easy to use; however, they are relatively inefficient, contain significant dead space in the tubing, and are time-consuming and expensive.45 A mask must be used in conjunction with a nebulizer; the use of blow-by (holding the tube or mask in front of the face) significantly reduces the pulmonary delivery of medication.45 Equivalent bronchodilation can be achieved when a beta-agonist is given by metered-dose inhaler (MDI) with a spacer and holding chamber.8,11,46-48 MDIs should never be used without an appropriate spacer device; an appropriate spacer maximizes drug delivery to the lungs while minimizing oropharyngeal deposition and swallowed drug, and therefore, side effects.45 Spacer devices for infants and young children should include masks, since they are nasal breathers.45
• Consider steroid therapy early for children with conditions known to benefit from their use, including asthma and BPD. Oral administration of prednisone is equally efficacious to parenteral administration, and is preferred.8,11 The onset of action is typically greater than four hours. For children who receive corticosteroids in the ED and will be discharged home, a course of 3-10 days of oral steroids at discharge is useful.8 Corticosteroids have not been shown to be beneficial in the treatment of bronchiolitis. (See Table 6.) Reassess frequently; a slowing respiratory rate in a tachypneic infant may represent improvement, or may be an ominous sign that the infant is tiring and respiratory failure is imminent. For children with severe intrathoracic airway narrowing or obstruction, treatment initially may result in increased wheezing as air exchange improves. With continued improvement, wheezing is heard later and later in expiration until the airways are dilated enough that no wheezing is heard.
• Provide a discharge plan for children sent home from the ED. The discharge plan should include instructions on frequency of beta-agonist therapy, and dose and duration of oral steroids or other medications recommended. The written plan also should include indications for returning to the ED if needed. Discharge teaching must include instruction and demonstration of the proper use of inhaler and spacer devices.
• Recommend or arrange follow-up with a primary care medical home within 3-5 days. A primary care provider is best suited to coordinate a work-up if the diagnosis is in question, and to follow the child for recurrent episodes of wheezing. Ongoing education and supervision for children who require chronic medications and treatment plans also are provided best in the primary care setting.
• Avoid the following therapies, which have been employed in the past but are not currently recommended:
Management—Specific Entities
Because of the overwhelming predominance of asthma and bronchiolitis as causes of wheezing in this age group, additional management guidelines for these entities follow.
Bronchiolitis. Provide cardiopulmonary monitoring as needed. Monitoring for apneic and bradycardic spells may be necessary during the acute stage of illness, particularly in very young infants (younger than 6 weeks of age) or infants with underlying cardiopulmonary diseases.
Consider a trial of bronchodilator therapy. Although studies have shown variable degrees of response of children with bronchiolitis to bronchodilator therapy, the response generally has not been clinically significant.51-53 Nebulized epinephrine has been shown to be more beneficial than albuterol in some studies.51,54 A one-dose trial of albuterol or epinephrine is reasonable, provided the child is evaluated immediately before and after administration. A response should be evident within 15-30 minutes; if there is not a clear, significant benefit, the dose should not be repeated. Deterioration associated with inhalation therapies for bronchiolitis is well-described; infants should be observed closely.50 (See Table 6.)
Provide supportive care and hospitalize infants with significant respiratory distress or who are unable to maintain oxygenation or hydration.
The following therapies are controversial or are not recommended:
• Corticosteroid use is controversial; most studies have failed to demonstrate benefit, and they are not currently recommended for previously healthy infants with RSV bronchiolitis.55 Studies that have shown benefit have not excluded infants with probable asthma, and corticosteroid therapy may be beneficial in the subset of infants with bronchiolitis who are predisposed to developing asthma.56-58 Further study is needed to clarify the use of corticosteroids for this disease.
• Antibiotics rarely are indicated, as associated bacterial pneumonia is very uncommon.55 Appropriate antibiotic therapy is warranted for infants with concomitant otitis media.
• Ribavirin, an antiviral agent with in vitro activity against RSV, is controversial, and generally not recommended because of its high cost, difficult administration, potential toxicity to health care providers, and questionable efficacy.55
Asthma. Provide steroids early. Steroids have been shown to speed recovery and reduce the likelihood of recurrence. Early use of oral steroids particularly is important in young children, as well as for children with moderate to severe exacerbations, and patients who will be admitted to the hospital.8 For patients taking oral steroids long-term, supplemental doses of steroids should be given, even if the current exacerbation is mild.8
Consider the addition of anticholinergic therapy to albuterol nebulizer therapy. Ipratropium bromide has been shown to improve lung function and decrease hospitalization in some, though not all, studies.8,11 Benefit is most evident in children with severe exacerbations of asthma and is less well-documented in moderate disease or in children younger than 2 years.59-61 (See Table 6.)
Hospitalize children in whom respiratory distress or low oxygen saturation persist despite therapy with bronchodilators, steroids, and anticholinergic medication. Children with risk factors for asthma-related death should be managed especially cautiously.8 (See Table 7.)
|
Recommend long-term control therapies for children with persistent or frequently recurring asthma symptoms. Long-term control therapy with inhaled corticosteroids is recommended for even very young children if wheezing is frequent, severe, or associated with risk factors for asthma. Children who are already on long-term control medications should be encouraged to continue them during their exacerbation. Although control therapies most often are prescribed and monitored by primary care physicians, the emergency medicine physician must be familiar with the standards of current asthma care.
Outcomes
The parent of a wheezing child often will ask whether the child has asthma. While asthma is the single most common cause of wheezing in the first 3 years of life, half or more of children who wheeze do not go on to have asthma.62,63 Defining which children who wheeze are at risk for persistent asthma could allow for better management and, potentially, for reduced morbidity and mortality.10
Children with recurrent wheezing in the first years of life who do not have an underlying anatomic abnormality or disease generally can be divided into two groups: those with transient early wheezing and those with atopic asthma. Transient early wheezing typically resolves by age 3 and generally is not associated with a family history of asthma or allergic sensitization.10
Although there is not a specific test or single clinical indicator to identify asthma, a number of risk factors and associations have been identified which help to define the subset of wheezing infants who most likely will become children with asthma. The most significant risk factors seem to be eczema in the patient or asthma in a parent; other risk factors and associations include allergic rhinitis in the first year of life, close family members with other atopic disease, recurrence of wheezing without an associated URI, perinatal passive smoke exposure, eosinophilia greater than 4%, elevated serum IgE level at 9 months of age, and positive allergy skin testing.9,10,35,37
The role of RSV infection during infancy as a risk factor for asthma is unclear. Infants who have had RSV infection tend to have an increased risk of recurrent wheezing during childhood, but their wheezing tends to decrease with age and often no longer is significant by adolescence; RSV infection alone does not appear to be a risk factor for atopic asthma.10,64
Summary
Wheezing is common in children during the first years of life. While the differential diagnosis is broad, a significant number of children with wheezing have asthma; a substantial proportion may have viral bronchiolitis or virally-mediated wheezing that resolves as the child ages.
Understanding the basic mechanisms of wheezing and using a systematic approach to diagnosis helps to institute the proper treatment in an efficient manner. Although bronchiolitis and asthma predominate, maintain a high index of suspicion for the “mimics of asthma” in infants and young children.
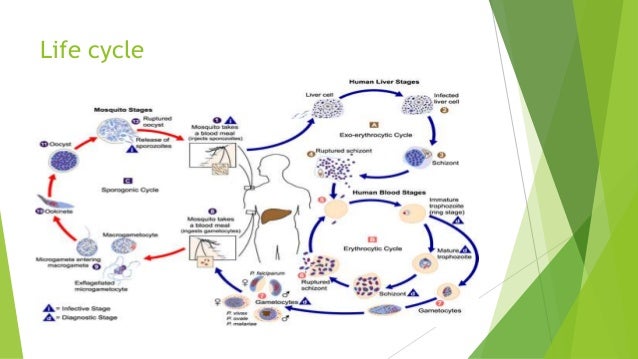
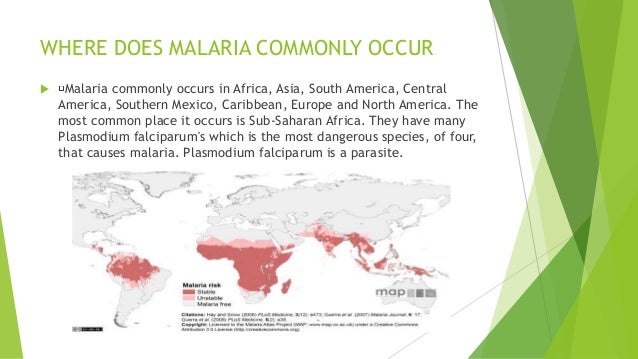
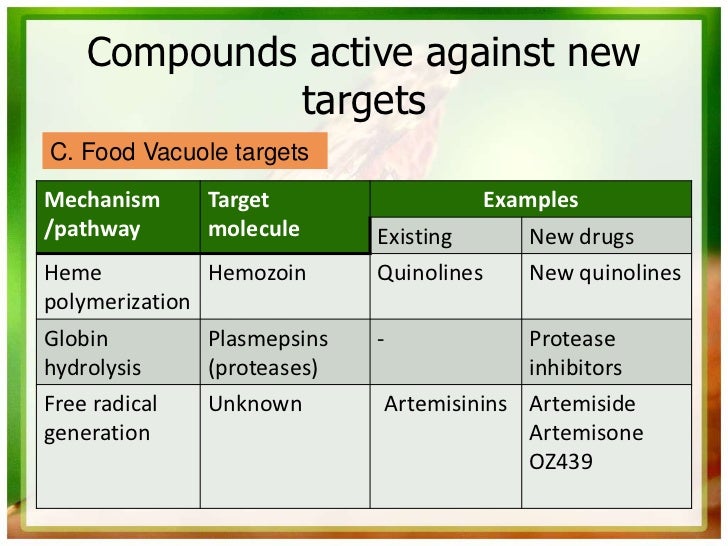
P. falciparum has also developed resistance to nearly all of the other currently available antimalarial drugs, such as sulfadoxine/pyrimethamine, mefloquine, halofantrine, and quinine.


















































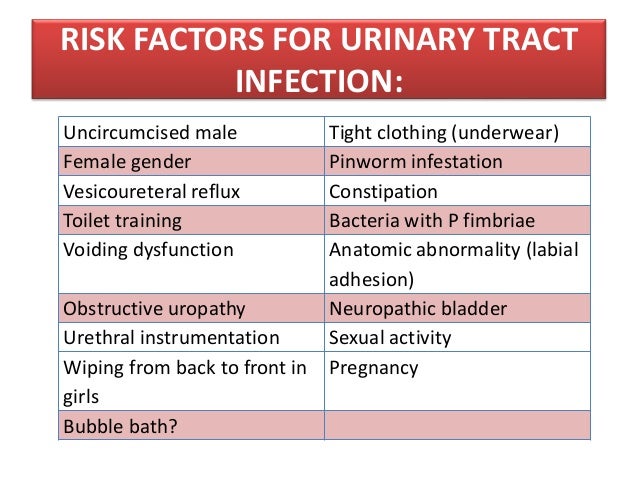
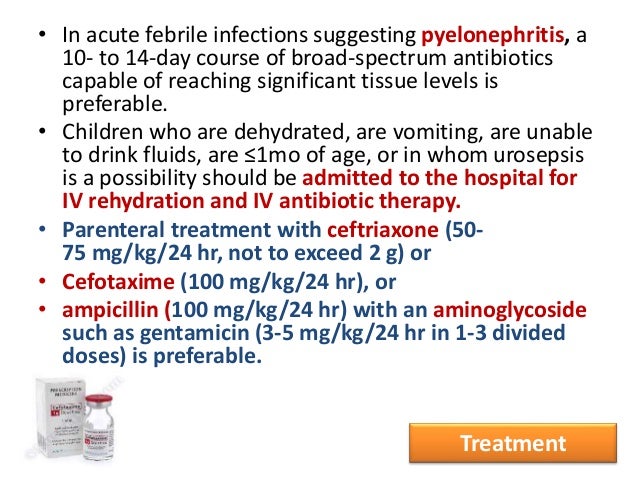
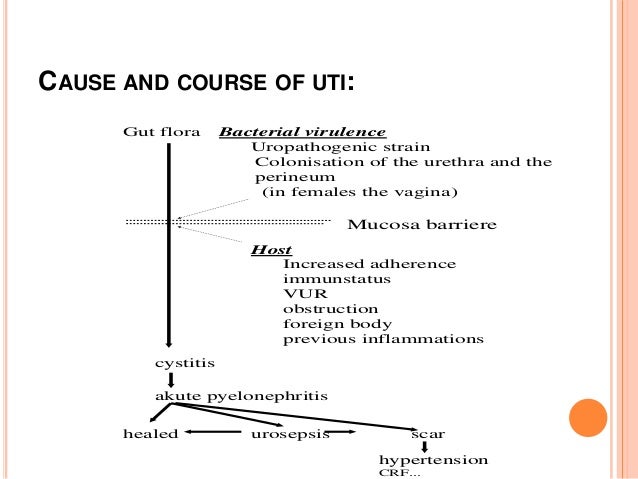
Acute urinary tract infections are relatively common in children, with 8 percent of girls and 2 percent of boys having at least one episode by seven years of age.
The most common pathogen is Escherichia coli, accounting for approximately 85 percent of urinary tract infections in children.
Renal parenchymal defects are present in 3 to 15 percent of children within one to two years of their first diagnosed urinary tract infection.
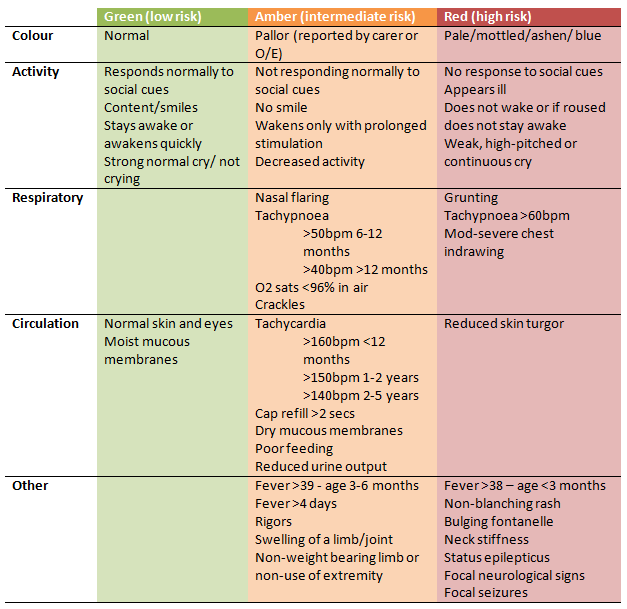
Clinical signs and symptoms of a urinary tract infection depend on the age of the child, but all febrile children two to 24 months of age with no obvious cause of infection should be evaluated for urinary tract infection (with the exception of circumcised boys older than 12 months).
Evaluation of older children may depend on the clinical presentation and symptoms that point toward a urinary source (e.g., leukocyte esterase or nitrite present on dipstick testing; pyuria of at least 10 white blood cells per high-power field and bacteriuria on microscopy).
Increased rates of E. coli resistance have made amoxicillin a less acceptable choice for treatment, and studies have found higher cure rates with trimethoprim/sulfamethoxazole. Other treatment options include amoxicillin/clavulanate and cephalosporins.
Prophylactic antibiotics do not reduce the risk of subsequent urinary tract infections, even in children with mild to moderate vesicoureteral reflux.
Constipation should be avoided to help prevent urinary tract infections. Ultrasonography, cystography, and a renal cortical scan should be considered in children with urinary tract infections.































 *************************************************************************************
*************************************************************************************






************************************************************************
BRETT WHITE, MD, Oregon Health and Science University, Portland, Oregon
Acute urinary tract infections are relatively common in children, with 8 percent of girls and 2 percent of boys having at least one episode by seven years of age. The most common pathogen is Escherichia coli, accounting for approximately 85 percent of urinary tract infections in children.
Renal parenchymal defects are present in 3 to 15 percent of children within one to two years of their first diagnosed urinary tract infection. Clinical signs and symptoms of a urinary tract infection depend on the age of the child, but all febrile children two to 24 months of age with no obvious cause of infection should be evaluated for urinary tract infection (with the exception of circumcised boys older than 12 months).
Evaluation of older children may depend on the clinical presentation and symptoms that point toward a urinary source (e.g., leukocyte esterase or nitrite present on dipstick testing; pyuria of at least 10 white blood cells per high-power field and bacteriuria on microscopy).
Increased rates of E. coli resistance have made amoxicillin a less acceptable choice for treatment, and studies have found higher cure rates with trimethoprim/sulfamethoxazole.
Other treatment options include amoxicillin/clavulanate and cephalosporins.
Prophylactic antibiotics do not reduce the risk of subsequent urinary tract infections, even in children with mild to moderate vesicoureteral reflux. Constipation should be avoided to help prevent urinary tract infections. Ultrasonography, cystography, and a renal cortical scan should be considered in children with urinary tract infections.
Guidelines regarding the diagnosis, treatment, and follow-up of urinary tract infections (UTIs) in children continue to evolve. Although a somewhat less aggressive approach to evaluation is now recommended, it is important for primary care physicians to appropriately diagnose and treat UTIs in children. Some underlying etiologies, including renal scarring and renal disease, can lead to considerable morbidity later in life.
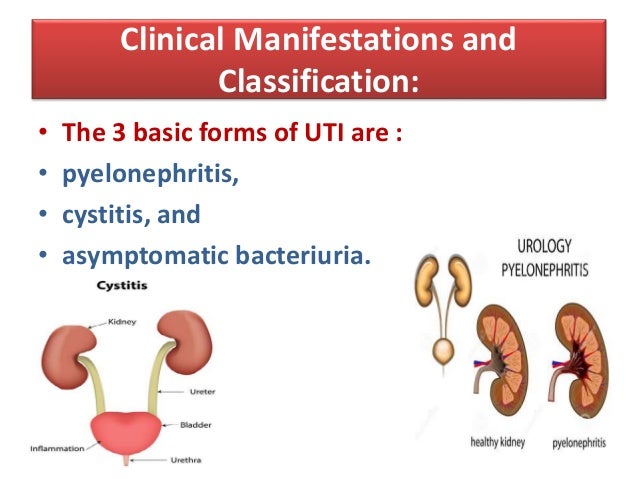
Acute UTIs are relatively common in children. By seven years of age, 8 percent of girls and 2 percent of boys will have at least one episode.1 In a study of infants presenting to pediatric emergency departments, the prevalence of UTI in infants younger than 60 days with a temperature greater than 100.4°F (38°C) was 9 percent.2 The reference standard for the diagnosis of UTI is a single organism cultured from a specimen obtained at the following concentrations: suprapubic aspiration specimen, greater than 1,000 colony-forming units per mL; catheter specimen, greater than 10,000 colony-forming units per mL; or clean-catch, midstream specimen, 100,000 colony-forming units per mL or greater.3–5 Use of lower colony counts in symptomatic patients has been advocated, 6 although this has not been included in established guidelines.
Common uropathogens include Escherichia coli (accounting for approximately 85 percent of UTIs in children), Klebsiella, Proteus, Enterobacter, Citrobacter, Staphylococcus saprophyticus, and Enterococcus.7 A systematic review found that renal parenchymal defects are identified in 3 to 15 percent of children within one to two years of their first diagnosed UTI.8 Long-term complications of UTI associated with renal scarring include hypertension, chronic renal failure, and toxemia in pregnancy. Long-term follow-up data are limited, although one Swedish study found that among patients who had renal scarring from pyelonephritis during childhood, 23 percent developed hypertension and 10 percent developed end-stage renal disease.9 However, more recent studies question the association between pyelonephritis and end-stage renal disease.10 Baseline abnormalities of the urogenital tract have been reported in up to 3.2 percent of healthy, screened infants.11 Additionally, obstructive anomalies are found in up to 4 percent and vesicoureteral reflux in 8 to 40 percent of children being evaluated for their first UTI. Children younger than two years may be at greater risk of parenchymal defects than older children.12
| Clinical recommendation | Evidence rating | References |
|---|---|---|
| UTI should be suspected in patients with leukocyte esterase and nitrite present on dipstick testing, or with pyuria of at least 10 white blood cells per high-power field and bacteriuria on microscopy. | 1316 | |
| In young children, urine samples collected with a bag are unreliable in the evaluation of UTI. | 17 | |
| The recommended initial antibiotic for most children with UTI is trimethoprim/sulfamethoxazole (Bactrim, Septra). Alternative antibiotics include amoxicillin/clavulanate (Augmentin) or cephalosporins, such as cefixime (Suprax), cefpodoxime, cefprozil (Cefzil), or cephalexin (Keflex). | 10 | |
| A two- to four-day course of oral antibiotics is as effective as a seven- to 14-day course in children with a lower UTI. A single-dose or single-day course is not recommended. | 1921 | |
| Children with acute pyelonephritis can be treated effectively with oral antibiotics (e.g., amoxicillin/clavulanate, cefixime, ceftibuten [Cedax]) for 10 to 14 days or with short courses (two to four days) of intravenous therapy followed by oral therapy. | 24 | |
| Prophylactic antibiotics do not reduce the risk of recurrent UTIs, even in children with mild to moderate vesicoureteral reflux. | 2527 | |
| Routine circumcision in boys does not reduce the risk of UTI enough to justify the risk of surgical complications. | 32 |
UTI = urinary tract infection.
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.xml.
Clinical signs and symptoms of a UTI depend on the age of the child. Newborns with UTI may present with jaundice, sepsis, failure to thrive, vomiting, or fever. In infants and young children, typical signs and symptoms include fever, strong-smelling urine, hematuria, abdominal or flank pain, and new-onset urinary incontinence. School-aged children may have symptoms similar to adults, including dysuria, frequency, or urgency. Boys are at increased risk of UTI if younger than six months, or if younger than 12 months and uncircumcised. Girls are generally at an increased risk of UTI, particularly if younger than one year.3 Physical examination findings can be nonspecific but may include suprapubic tenderness or costovertebral angle tenderness.
Dipstick tests for UTI include leukocyte esterase, nitrite, blood, and protein. Leukocyte esterase is the most sensitive single test in children with a suspected UTI. The test for nitrite is more specific but less sensitive. A negative leukocyte esterase result greatly reduces the likelihood of UTI, whereas a positive nitrite result makes it much more likely; the converse is not true, however. Dipstick tests for blood and protein have poor sensitivity and specificity in the detection of UTI and may be misleading. Accuracy of positive findings is as follows (assumes a 10 percent pretest probability)13:
All febrile children between two and 24 months of age with no obvious cause of infection should be evaluated for UTI, with the exception of circumcised boys older than 12 months.14 Older children should be evaluated if the clinical presentation points toward a urinary source. The National Institute for Health and Clinical Excellence in the United Kingdom endorses incorporating specific strategies for urine testing based on the child’s age (Figure 1).15 In this model, microscopy and urine culture should be performed in children younger than three years instead of dipstick testing. The presence of pyuria of at least 10 white blood cells per high-power field and bacteriuria are recommended as the criteria for diagnosing UTI with microscopy.16 In young children, urine samples collected with a bag are unreliable compared with samples collected with a catheter.17 Therefore, in a child who is unable to provide a clean-catch specimen, catheterization should be considered. If urine cannot be cultured within four hours of collection, the sample should be refrigerated.
Imaging procedures with the highest ratings from the American College of Radiology Appropriateness Criteria for further evaluation of select children with UTIs are renal and bladder ultrasonography, radionuclide cystography or voiding cystourethrography, and renal cortical scan.18 Renal and bladder ultrasonography is effective for evaluating anatomy, but is unreliable for detecting vesicoureteral reflux. Radionuclide cystography or voiding cystourethrography is effective for screening and grading vesicoureteral reflux, but involves radiation exposure and catheterization. Although voiding cystourethrography is suggested for either girls or boys, radionuclide cystography is suggested only for girls because voiding cystourethrography is needed for adequate anatomic imaging of the urethra and bladder in boys. A renal cortical scan (also called scintigraphy or DMSA scan) uses technetium and is effective for assessing renal scarring, but requires intravenous injection of radioisotope.
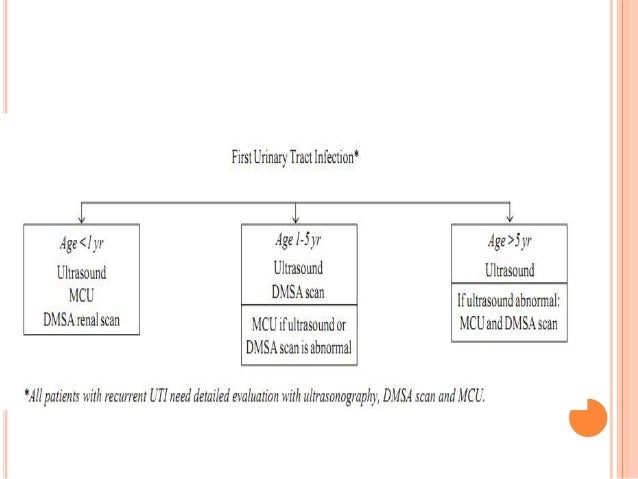
Long-term outcome studies have not been performed to determine the best initial imaging study in children diagnosed with UTI. Guidelines based on observational studies and expert opinion recommend that all boys, girls younger than three years, and girls three to seven years of age with a temperature of 101.3°F (38.5°C) or greater receive cystography and ultrasonography with a first-time UTI.3 An optional imaging strategy for febrile children with UTI, especially those older than three years, is to first perform ultrasonography and a renal cortical scan. This strategy avoids bladder catheterization with cystography and minimizes radiation exposure if the results of the scan are normal. However, if pyelonephritis or cortical scarring is found on the renal cortical scan, cystography is indicated.3
Observation without imaging should be considered in girls three years or older with a temperature less than 101.3° F and in all girls older than seven years.3 The family should share in the decision to perform imaging with the first UTI or delay imaging until the second UTI, if it occurs. Figure 2 is an algorithm for imaging strategies in children with UTI.3
Although fever may be the sole presenting symptom in children younger than 24 months, physical examination findings may point toward an alternative diagnosis, including otitis media, gastroenteritis, or upper respiratory tract infection. Occult bacteremia should always be considered, although the probability of this diagnosis is much lower than UTI (less than 1 versus 7 percent) in fully immunized children with no other identifiable potential source for fever on physical examination.4 Urinary calculi, urethritis (including a sexually transmitted infection), dysfunctional elimination, and diabetes mellitus must be considered in verbal children with urinary tract problems.
Although amoxicillin has traditionally been a first-line antibiotic for UTI, increased rates of E. coli resistance have made it a less acceptable choice, and studies have found higher cure rates with trimethoprim/sulfamethoxazole (Bactrim, Septra). Other choices include amoxicillin/clavulanate (Augmentin) or cephalosporins, such as cefixime (Suprax), cefpodoxime, cefprozil (Cefzil), or cephalexin (Keflex).10 Table 1 lists commonly used antibiotics, with dosing information and adverse effects. Physicians should be aware of local bacterial resistance patterns that might affect antibiotic choices.
| Antibiotic | Dosing | Common adverse effects |
|---|---|---|
| Amoxicillin/clavulanate (Augmentin) | 25 to 45 mg per kg per day, divided every 12 hours | Diarrhea, nausea/vomiting, rash |
| Cefixime (Suprax) | 8 mg per kg every 24 hours or divided every 12 hours | Abdominal pain, diarrhea, flatulence, rash |
| Cefpodoxime | 10 mg per kg per day, divided every 12 hours | Abdominal pain, diarrhea, nausea, rash |
| Cefprozil (Cefzil) | 30 mg per kg per day, divided every 12 hours | Abdominal pain, diarrhea, elevated results on liver function tests, nausea |
| Cephalexin (Keflex) | 25 to 50 mg per kg per day, divided every 6 to 12 hours | Diarrhea, headache, nausea/vomiting, rash |
| Trimethoprim/sulfamethoxazole (Bactrim, Septra) | 8 to 10 mg per kg per day, divided every 12 hours | Diarrhea, nausea/vomiting, photosensitivity, rash |
A Cochrane review analyzing short-duration (two to four days) versus standard-duration (seven to 14 days) oral antibiotics in 652 children with lower UTIs found no significant difference in positive urine cultures between the therapies immediately after treatment (eight studies: relative risk = 1.06; 95% confidence interval, 0.64 to 1.76) or 15 months after treatment (10 studies: relative risk = 0.95; 95% confidence interval, 0.70 to 1.29). There was also no significant difference between short- and standard-duration therapies in the development of resistant organisms at the end of treatment.19 Thus, a two- to four-day course of oral antibiotics appears to be as effective as a seven- to 14-day course in children with lower UTIs.19 A single-dose or single-day course may be less effective than longer courses of oral antibiotics and is not recommended.2021
When the presenting symptoms are nonspecific for a UTI or the urine dipstick test is nondiagnostic, there may be a delay in treatment while culture results are pending. Parents can be reassured that antibiotics initiated 24 hours after the onset of fever are not associated with a higher risk of parenchymal defects than immediate antibiotics in children younger than two years.22 However, delaying antibiotics by four days or more may increase the risk of renal scarring.8
Fluoroquinolones are not usually used in children because of potential concerns about sustained injury to developing joints, although there is no compelling evidence supporting the occurrence of this phenomenon. Fluoroquinolones may be useful when infection is caused by multidrug-resistant pathogens for which there is no safe and effective alternative, parenteral therapy is not feasible, and no other effective oral agent is available. Guidelines from the American Academy of Pediatrics recommend limiting fluoroquinolone therapy to patients with UTIs caused by Pseudomonas aeruginosa or other multidrug-resistant, gram-negative bacteria.23 Ciprofloxacin (Cipro) is approved by the U.S. Food and Drug Administration for complicated UTIs and pyelonephritis attributable to E. coli in patients one to 17 years of age.23
A Cochrane review concluded that children with acute pyelonephritis can be treated effectively with oral antibiotics (e.g., amoxicillin/clavulanate, cefixime, ceftibuten [Cedax]) for 10 to 14 days or with short-courses (two to four days) of intravenous therapy followed by oral therapy. If intravenous therapy is used, single daily dosing with aminoglycosides is safe and effective. Studies are needed to determine the optimal duration of intravenous therapy in children with acute pyelonephritis, but 10 to 14 days is typical.24 Hospitalization should be considered for any child that is unable to tolerate oral intake or when the diagnosis is uncertain in a markedly ill child.
Follow-up assessment to confirm an appropriate clinical response should be performed 48 to 72 hours after initiating antimicrobial therapy in all children with UTI. Culture and susceptibility results may indicate that a change of antibiotic is necessary. If expected clinical improvement does not occur, consider further evaluation (e.g., laboratory studies, imaging, consultation with subspecialists). Referral to a subspecialist is indicated if vesicoureteral reflux, renal scarring, anatomic abnormalities, or renal calculi are discovered, or if invasive imaging procedures are considered.
In an observational study of otherwise healthy children with a first UTI, antibiotic prophylaxis was not associated with a reduced risk of recurrent UTI and increased the risk of treatment-resistant pathogens.25 A randomized controlled trial of children two months to seven years of age found that prophylactic antibiotics for 12 months following a febrile UTI did not reduce the risk of subsequent UTI, even in children with mild to moderate vesicoureteral reflux.26 Another randomized controlled trial of children and adolescents with pyelonephritis found that antibiotic prophylaxis did not prevent subsequent UTIs in patients with no documented vesicoureteral reflux or with mild to moderate vesicoureteral reflux.27 Antibiotic prophylaxis may be more beneficial in children with more severe vesicoureteral reflux, however.28 The most recent Cochrane review on the subject concluded that large, properly randomized, double-blind studies are needed to determine the effectiveness of long-term antibiotics for the prevention of UTI in susceptible children.1 Additionally, continuous antibiotic prophylaxis in children younger than two and a half years with vesicoureteral reflux may not decrease the risk of pyelonephritis or renal damage.29
Constipation should be addressed in infants and children who have had a UTI to help prevent subsequent infections.30 There is some evidence that cranberry juice decreases symptomatic UTIs over 12-months, particularly in women with recurrent UTIs.31 The effectiveness of cranberry juice in children is less certain, and the high dropout rate in studies indicates that cranberry juice may not be acceptable for long-term prevention. A systematic review concluded that routine circumcision in boys does not reduce the risk of UTI enough to justify the risk of surgical complications.
Findings indicate that CSE is linked to developmental impairments within six weeks of the event, and that the impairments persisted at the one-year follow-up.
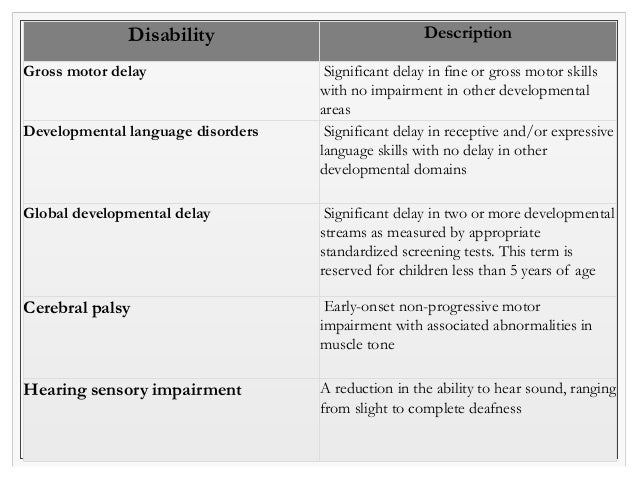
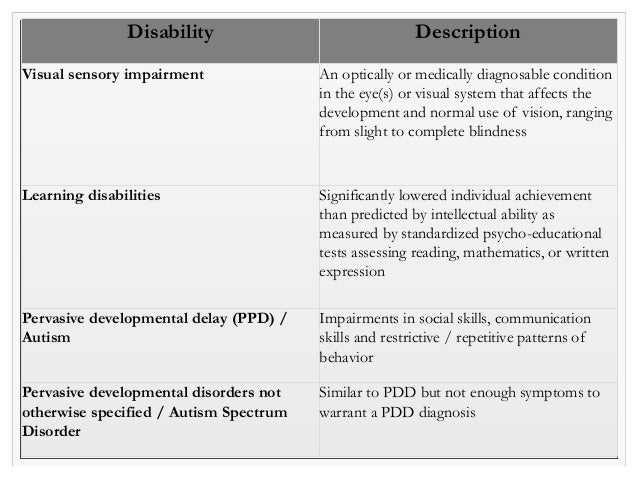
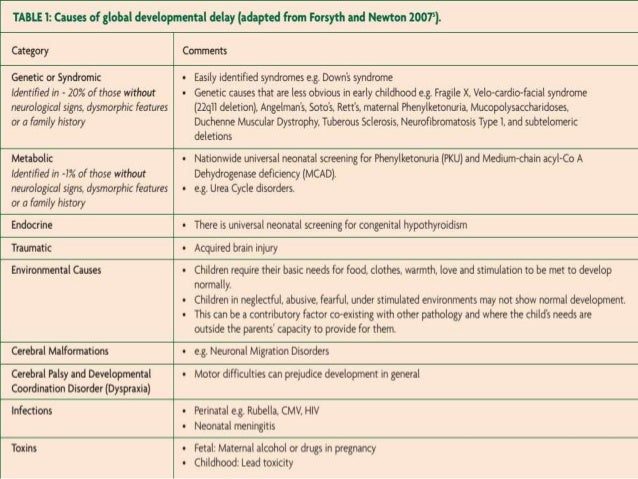
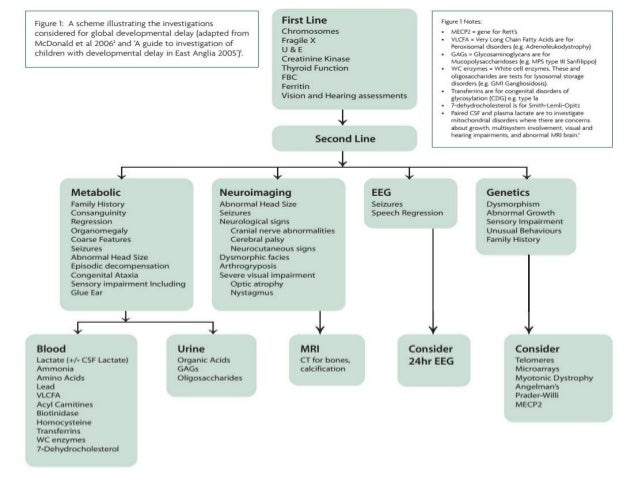
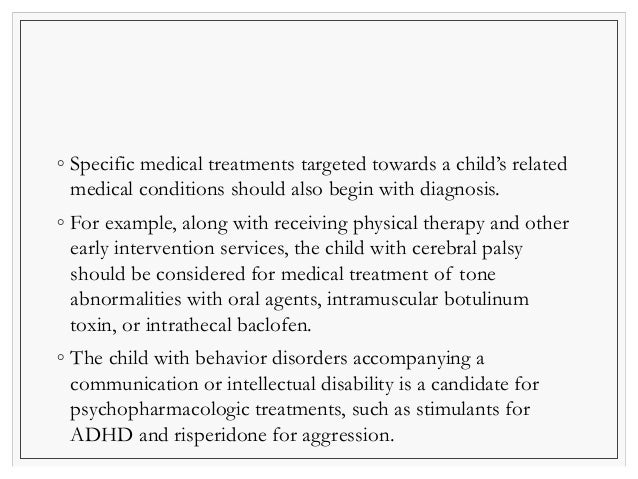
Microcephaly, seizures, and developmental delay (MCSZ) is a condition characterized by an abnormally small head size (microcephaly) and neurological problems related to impaired brain development before birth.
Affected individuals typically have recurrent seizures (epilepsy) beginning in infancy and delayed development of motor skills, such as sitting and walking. Speech is also delayed, and some affected individuals are never able to speak.
Intellectual disability and behavior problems, primarily hyperactivity, are also common features of MCSZ.
This is a new mental retardation syndrome that should be considered in children with recurrent febrile convulsions, developmental delay, and obesity.
Researchers have determined that developmental delays are present in children within six weeks following convulsive status epilepticus (CSE)—a seizure lasting longer than thirty minutes.







 [
[






























*******************************************************************































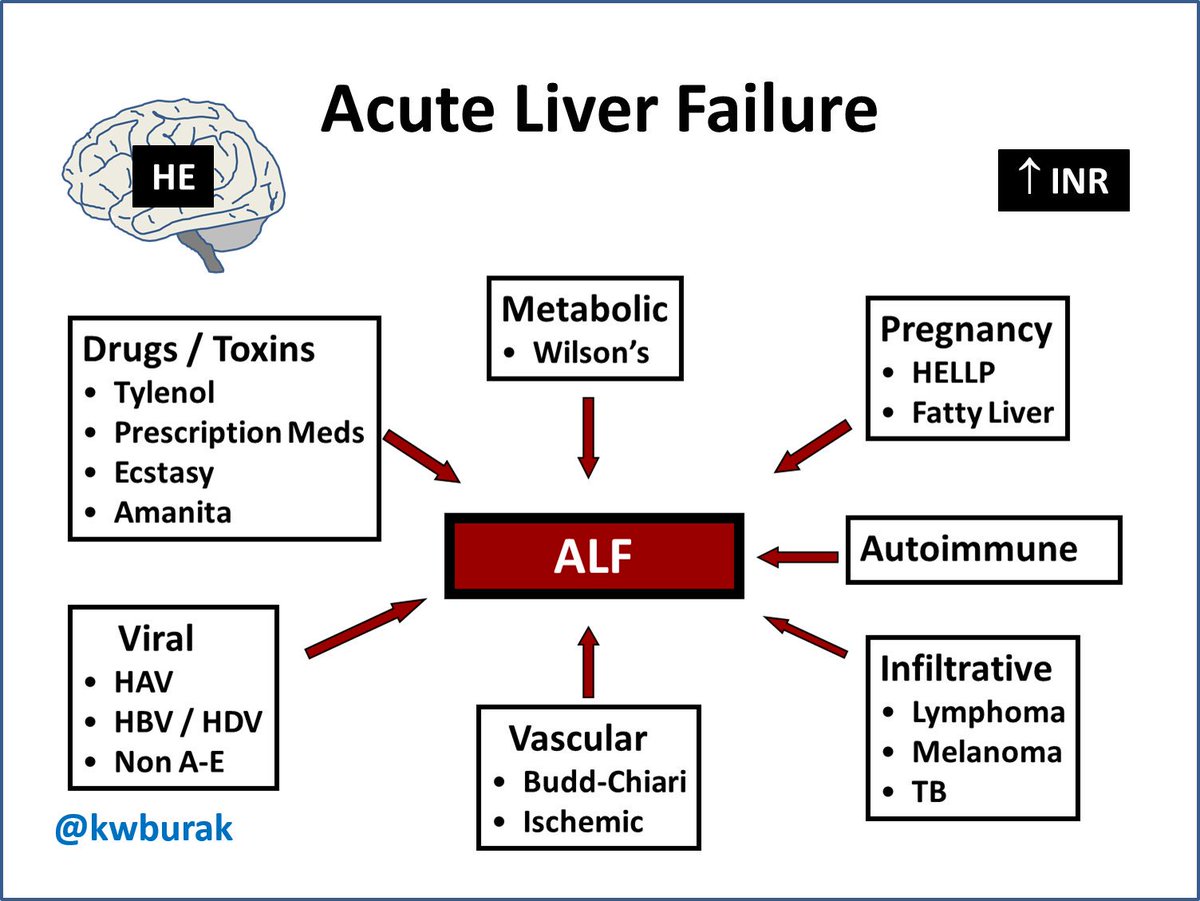
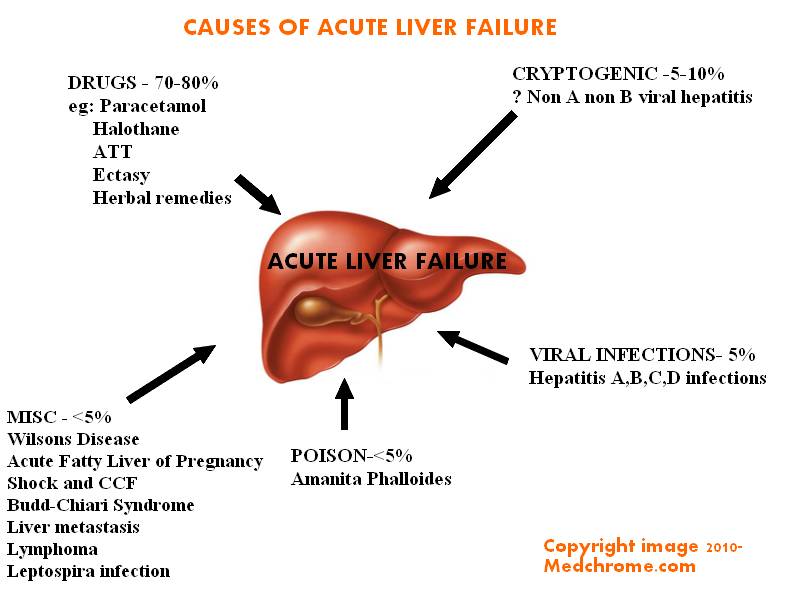
The most common causes of acute liver failure are viral hepatitis (in developing countries) and drugs and toxins (in developed countries).
Acute liver failure is characterized by jaundice, coagulopathy, and encephalopathy.
Confirm the diagnosis by finding prolongation of PT or clinical manifestations of encephalopathy in patients with hyperbilirubinemia and elevated aminotransferase levels.
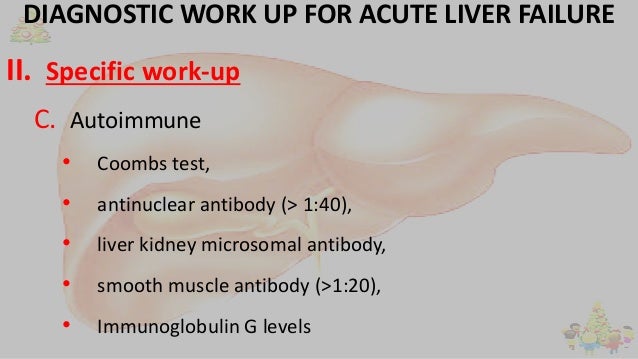
Determine the cause by assessing history of drug use and exposure to toxins and doing hepatitis virus serologic tests, autoimmune markers, and other tests based on clinical suspicion.
Treat complications intensively, usually in an ICU.
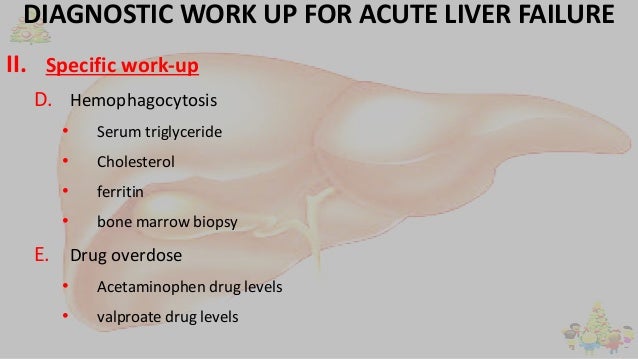
Consider N-acetylcysteine for acetaminophen-induced liver failure and liver transplantation for patients with poor prognostic factors (eg, age < 10 or > 40, severe encephalopathy, severe prolongation of PT, idiosyncratic drug reaction, Wilson disease).













































********************************************************************************
Liver failure can be classified in several ways, but no system is universally accepted (see table Classification of Liver Failure).
Overall, the most common causes of acute liver failure are
Viruses, primarily hepatitis B
Drugs and toxins, most commonly acetaminophen
In developing countries, viral hepatitis is usually considered the most common cause; in developed countries, toxins are usually considered the most common cause.
Overall, the most common viral cause is hepatitis B, often with hepatitis D coinfection; hepatitis C is not a common cause.
Other possible viral causes include cytomegalovirus, Epstein-Barr virus, herpes simplex virus, human herpesvirus 6, parvovirus B19, varicella-zoster virus, hepatitis A virus (rarely), hepatitis E virus (especially if contracted during pregnancy), and viruses that cause hemorrhagic fever (see Overview of Arbovirus, Arenavirus, and Filovirus Infections).
The most common toxin is acetaminophen; toxicity is dose-related. Predisposing factors for acetaminophen-induced liver failure include preexisting liver disease, chronic alcohol use, and use of drugs that induce the cytochrome P-450 enzyme system (eg, anticonvulsants). Other toxins include amoxicillin/clavulanate, halothane, iron compounds, isoniazid, nonsteroidal anti-inflammatory drugs (NSAIDs), some compounds in herbal products, and Amanita phalloides mushrooms (see Liver Injury Caused by Drugs). Some drug reactions are idiosyncratic.
Less common causes include
Vascular disorders
Metabolic disorders
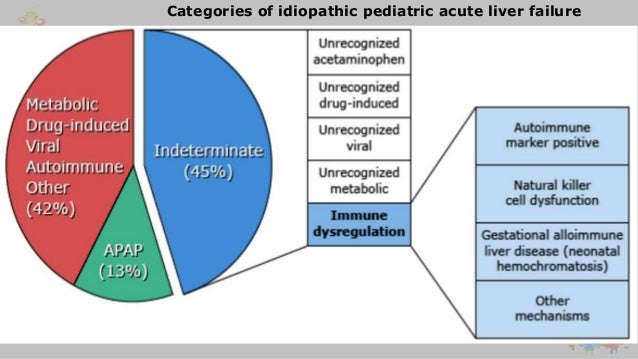
Vascular causes include hepatic vein thrombosis (Budd-Chiari syndrome), ischemic hepatitis, portal vein thrombosis, and hepatic sinusoidal obstruction syndrome (also called hepatic veno-occlusive disease), which is sometimes drug- or toxin-induced. Metabolic causes include acute fatty liver of pregnancy, HELLP syndrome (hemolysis, elevated values on liver tests, and low platelets), Reye syndrome, and Wilson disease. Other causes include autoimmune hepatitis, metastatic liver infiltration, heatstroke, and sepsis. The cause cannot be determined in up to 20% of cases.
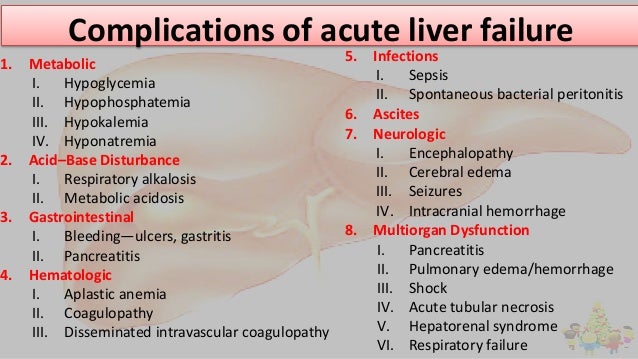
In acute liver failure, multiple organ systems malfunction, often for unknown reasons and by unknown mechanisms. Affected systems include
Hepatic: Hyperbilirubinemia is almost always present at presentation. The degree of hyperbilirubinemia is one indicator of the severity of liver failure. Coagulopathy due to impaired hepatic synthesis of coagulation factors is common. Hepatocellular necrosis, indicated by increased aminotransferase levels, is present.
Cardiovascular: Peripheral vascular resistance and blood pressure (BP) decrease, causing hyperdynamic circulation with increased heart rate and cardiac output.
Cerebral: Portosystemic encephalopathy occurs, possibly secondary to increased ammonia production by nitrogenous substances in the gut. Cerebral edema is common among patients with severe encephalopathy secondary to acute liver failure; uncal herniation is possible and usually fatal.
Renal: For unknown reasons, acute kidney injury occurs in up to 50% of patients. Because blood urea nitrogen (BUN) level depends on hepatic synthetic function, the level may be misleadingly low; thus, the creatinine level better indicates kidney injury. As in hepatorenal syndrome, urine sodium and fractional sodium excretion decrease even when diuretics are not used and tubular injury is absent (as may occur when acetaminophen toxicity is the cause).
Immunologic: Immune system defects develop; they include defective opsonization, deficient complement, and dysfunctional white blood cells (WBCs) and killer cells. Bacterial translocation from the gastrointestinal tract increases. Respiratory and urinary tract infections and sepsis are common; pathogens can be bacterial, viral, or fungal.
Metabolic: Both metabolic and respiratory alkalosis may occur early. If shock develops, metabolic acidosis can supervene. Hypokalemia is common, in part because sympathetic tone is decreased and diuretics are used. Hypophosphatemia and hypomagnesemia can develop. Hypoglycemia may occur because hepatic glycogen is depleted and gluconeogenesis and insulin degradation are impaired.
Pulmonary: Noncardiogenic pulmonary edema may develop.
Characteristic manifestations are altered mental status (usually part of portosystemic encephalopathy) and jaundice.
Manifestations of chronic liver disease such as ascites argue against the acuity of the condition but can be present in subacute liver failure.
Other symptoms may be nonspecific (eg, malaise, anorexia) or result from the causative disorder. Fetor hepaticus (a musty or sweet breath odor) and motor dysfunction are common. Tachycardia, tachypnea, and hypotension may occur with or without sepsis. Signs of cerebral edema can include obtundation, coma, bradycardia, and hypertension. Patients with infection sometimes have localizing symptoms (eg, cough, dysuria), but these symptoms may be absent. Despite prolonged international normalized ratio (INR) , bleeding is rare unless patients are in disseminated intravascular coagulation (DIC). This is because patients with acute liver failure have a re-balanced distribution of pro- and anticoagulant factors and, if anything, these patients are more frequently hypercoagulable (1,2).
Prolongation of prothrombin time (PT) and/or clinical manifestations of encephalopathy in patients with hyperbilirubinemia and elevated aminotransferase levels
To determine the cause: History of drug use, exposure to toxins, hepatitis virus serologic tests, autoimmune markers, and other tests based on clinical suspicion
Acute liver failure should be suspected if patients without underlying chronic liver disease or cirrhosis have acute onset of jaundice and/or elevated transaminases that is accompanied by coagulopathy and mental status changes. Patients with known liver disease who acutely decompensate are not considered to h
ave acute liver failure but rather acute-on-chronic liver failure, which has different pathophysiology from that of acute liver failure.
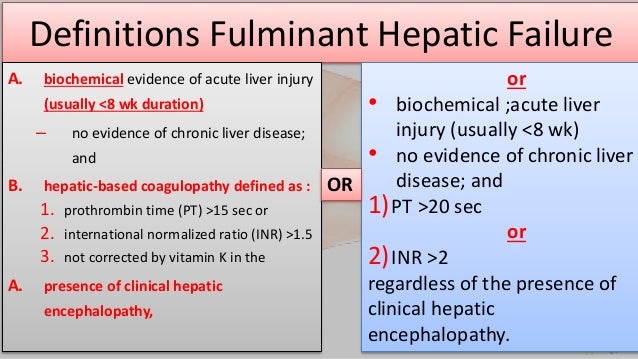
Laboratory tests to confirm the presence and severity of liver failure include liver enzyme and bilirubin levels and PT. Acute liver failure is usually considered confirmed if sensorium is altered or PT is prolonged by > 4 seconds or if INR is > 1.5 in patients who have clinical and/or laboratory evidence of acute liver injury. Evidence of cirrhosis suggests that liver failure is chronic.
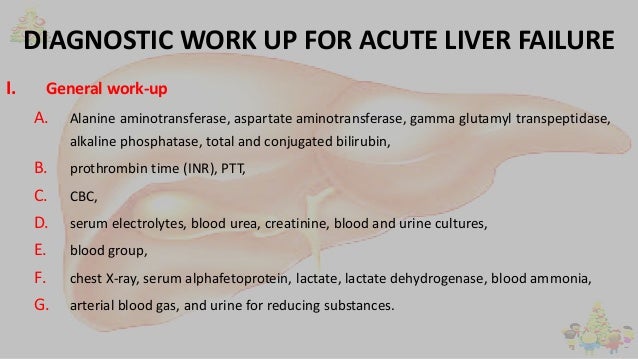
Patients with acute liver failure should be tested for complications. Tests usually done during the initial evaluation include complete blood count (CBC), serum electrolytes (including calcium, phosphate, and magnesium), renal function tests, and urinalysis. If acute liver failure is confirmed, arterial blood gases (ABGs), amylase and lipase, and blood type and screen should also be done.
Plasma ammonia is sometimes recommended for diagnosing encephalopathy or monitoring its severity. If patients have hyperdynamic circulation and tachypnea, cultures (blood, urine, ascitic fluid) and chest x-ray should be done to rule out infection. If patients have impaired or worsening mental status, particularly those with coagulopathy, head CT should be done to rule out cerebral edema or less likely intracranial bleeding.
To determine the cause of acute liver failure, clinicians should take a complete history of toxins ingested, including prescription and over-the-counter (OTC) drugs, herbal products, and dietary supplements. Tests done routinely to determine the cause include
Viral hepatitis serologic tests (eg, IgM antibody to hepatitis A virus [IgM anti-HAV], hepatitis B surface antigen [HBsAg], IgM antibody to hepatitis B core antigen [IgM anti-HBcAg], antibody to hepatitis C virus [anti-HCV])
Autoimmune markers (eg, antinuclear antibodies [ANA], anti–smooth muscle antibodies, immunoglobulin levels)
Other testing is done based on findings and clinical suspicion, as for the following:
Recent travel to developing countries: Tests for hepatitis A, B, D, and E
Females of child-bearing age: Pregnancy testing
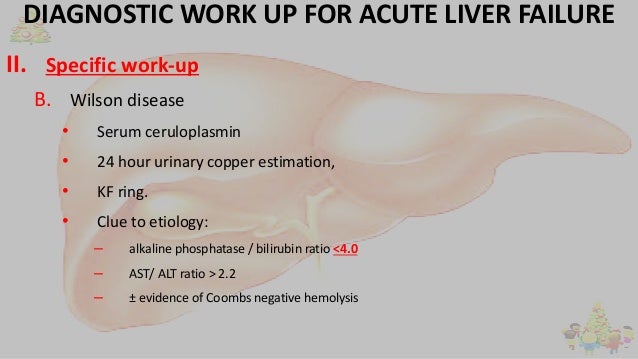
Age < 40 and relatively normal aminotransferase levels: Ceruloplasmin level to check for Wilson disease
Suspicion of a disorder with structural abnormalities (eg, Budd-Chiari syndrome, portal vein thrombosis, liver metastases): Ultrasonography and sometimes other imaging
Patients should be monitored closely for complications (eg, subtle changes in vital signs compatible with infection), and the threshold for testing should be low. For example, clinicians should not assume worsening mental status is due to encephalopathy; in such cases, head CT and often bedside glucose testing should be done. Routine laboratory testing (eg, daily PT, serum electrolytes, renal function tests, blood glucose, and ABGs) should be repeated frequently in most cases. However, testing may need to be more frequent (eg, blood glucose every 2 hours in patients with severe encephalopathy).
Prediction of prognosis can be difficult. Important predictive variables include
Degree of encephalopathy: Worse when encephalopathy is severe
Patient age: Worse when age is < 10 or > 40 years
PT: Worse when PT is prolonged
Various scores (for example, King’s College criteria or Acute Physiologic Assessment and Chronic Health Evaluation II [APACHE II] score) can predict prognosis in populations of patients but are not highly accurate for individual patients.
Supportive measures
N-Acetylcysteine for acetaminophen toxicity
Sometimes liver transplantation
(See also the American Association for the Study of Liver Diseases practice guideline Management of Acute Liver Failure: Update 2011.)
Whenever possible, patients should be treated in an intensive care unit at a center capable of liver transplantation. Patients should be transported as soon as possible because deterioration can be rapid and complications (eg, bleeding, aspiration, worsening shock) become more likely as liver failure progresses.
Intensive supportive therapy is the mainstay of treatment. Drugs that could worsen manifestations of acute liver failure (eg, hypotension, sedation) should be avoided or used in the lowest possible doses.
For hypotension and acute kidney injury, the goal of treatment is maximizing tissue perfusion. Treatment includes IV fluids and usually, until sepsis is excluded, empiric antibiotics. If hypotension is refractory to about 20 mL/kg of crystalloid solution, clinicians should consider measuring pulmonary capillary wedge pressure to guide fluid therapy. If hypotension persists despite adequate filling pressures, clinicians should consider using pressors (eg, dopamine, epinephrine, norepinephrine).
For encephalopathy, the head of the bed is elevated 30° to reduce risk of aspiration;
intubation should be considered early. When selecting drugs and drug doses, clinicians should aim to minimize sedation so that they can monitor the severity of encephalopathy. Propofol is the usual induction drug for intubation because it protects against intracranial hypertension and has a brief duration of action, allowing rapid recovery from sedation. There is no evidence that treatments such as lactulose or rifaximin help alleviate encephalopathy in acute liver failure, although they are useful in portosystemic encephalopathy. Also, lactulose can cause ileus and produce gas that distends the intestines, which can be problematic if laparotomy is needed (eg, for liver transplantation) (1). Measures are taken to avoid increasing intracranial pressure (ICP) and avoid decreasing cerebral perfusion pressure:
To avoid sudden increases in ICP: Stimuli that could trigger a Valsalva maneuver are avoided (eg, lidocaine is given before endotracheal suctioning to prevent the gag reflex).
To temporarily decrease cerebral blood flow: Mannitol (0.5 to 1 g/kg, repeated once or twice as needed) can be given to induce osmotic diuresis, and possibly brief hyperventilation can be used, particularly when herniation is suspected.
To monitor ICP: It is not clear whether or when the risks of ICP monitoring (eg, infection, bleeding) outweigh the benefits of being able to detect cerebral edema early and being able to use ICP to guide fluid and pressor therapy; some experts recommend such monitoring if encephalopathy is severe. However, no data indicate that ICP monitoring impacts mortality (2). Goals of treatment are an ICP of < 20 mm Hg and a cerebral perfusion pressure of > 50 mm Hg.
Seizures are treated with phenytoin; benzodiazepines are avoided or used only in low doses because they cause sedation.
Infection is treated with antibacterial and/or antifungal drugs; treatment is started as soon as patients show any sign of infection (eg, fever; localizing signs; deterioration of hemodynamics, mental status, or renal function). Because signs of infection overlap with those of acute liver failure, infection is likely to be overtreated pending culture results.
Electrolyte deficiencies may require supplementation with sodium, potassium, phosphate, or magnesium.
Hypoglycemia is treated with continuous glucose infusion (eg, 10% dextrose), and blood glucose should be monitored frequently because encephalopathy can mask the symptoms of hypoglycemia.
Coagulopathy is treated with fresh frozen plasma if bleeding occurs, if an invasive procedure is planned, or possibly if coagulopathy is severe (eg, international normalized ratio [INR] > 7). Fresh frozen plasma is otherwise avoided because it may result in volume overload and worsening of cerebral edema. Also, when fresh frozen plasma is used, clinicians cannot follow changes in PT, which are important because PT is an index of severity of acute liver failure and is thus sometimes a criterion for transplantation. Recombinant factor VII is sometimes used instead of or with fresh frozen plasma in patients with volume overload. Its role is evolving. H2 blockers may help prevent gastrointestinal (GI) bleeding.
Nutritional support may be necessary if patients cannot eat. Severe protein restriction is unnecessary; 60 g/day is recommended.
Acute acetaminophen toxicity is treated with N-acetylcysteine. Because chronic acetaminophen toxicity can be difficult to diagnose, use of N-acetylcysteine should be considered if no cause for acute liver failure is evident. Whether N-acetylcysteine has a slight beneficial effect on patients with acute liver failure due to other conditions is under study.
Liver transplantation results in average 1-year survival rates of about 80%. Transplantation is thus recommended if prognosis without transplantation is worse. However, prediction is difficult and scores, such as King’s College criteria and the APACHE II (Acute Physiologic Assessment and Chronic Health Evaluation II) score, are not sufficiently sensitive and specific to be used as the only criteria for transplantation; thus, they are used as adjuncts to clinical judgment (eg, based on risk factors).
Further information regarding acute liver failure can be found in the European Association for the Study of the Liver (EASL) guidelines.
The most common causes of acute liver failure are viral hepatitis (in developing countries) and drugs and toxins (in developed countries).
Acute liver failure is characterized by jaundice, coagulopathy, and encephalopathy.
Confirm the diagnosis by finding prolongation of PT or clinical manifestations of encephalopathy in patients with hyperbilirubinemia and elevated aminotransferase levels.
Determine the cause by assessing history of drug use and exposure to toxins and doing hepatitis virus serologic tests, autoimmune markers, and other tests based on clinical suspicion.
Treat complications intensively, usually in an ICU.
Consider N-acetylcysteine for acetaminophen-induced liver failure and liver transplantation for patients with poor prognostic factors (eg, age < 10 or > 40, severe encephalopathy, severe prolongation of PT, idiosyncratic drug reaction, Wilson disease).